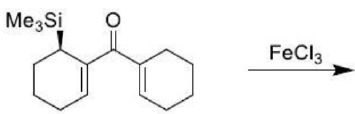
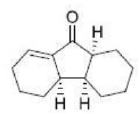
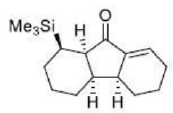
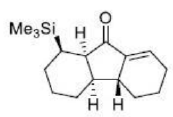
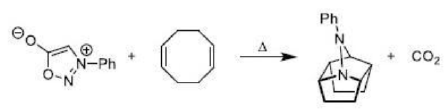
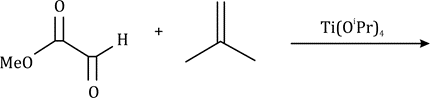
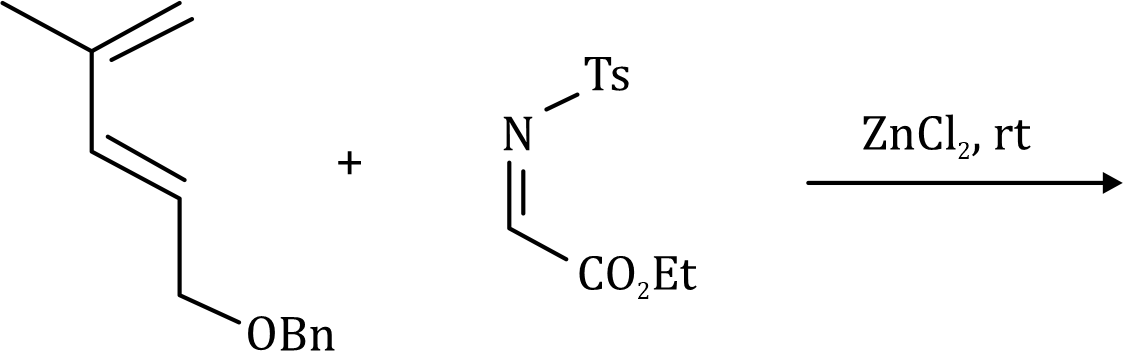
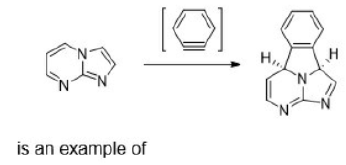
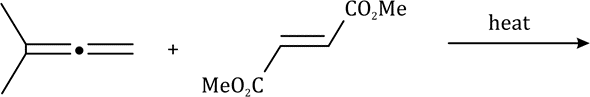Correct option is D
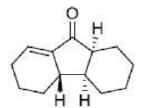
The Nazarov cyclization reaction is a chemical reaction used in organic chemistry for the synthesis of cyclopentenones.
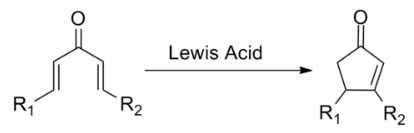
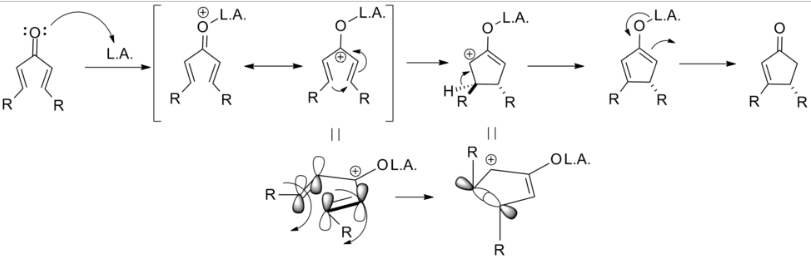
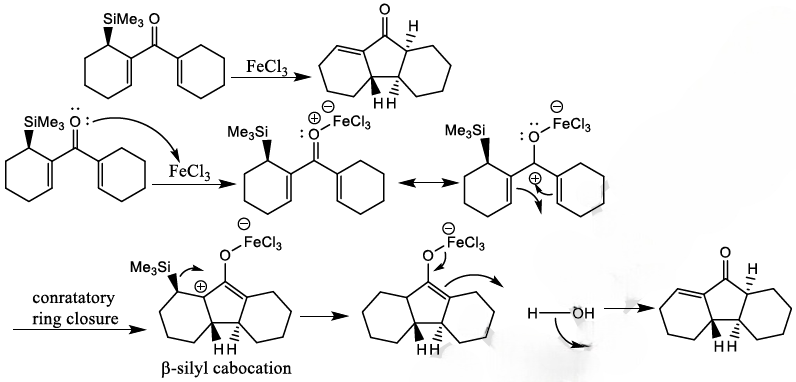
The Nazarov cyclization involves the activation of a divinyl ketone using a stoichiometric Lewis acid or protic acid promoter. The key step of the reaction mechanism involves a cationic 4π-electrocyclic ring closure which forms the cyclopentenone product.
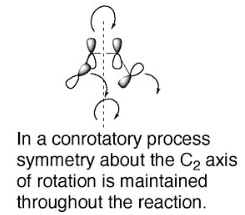
Activation of the ketone by the acid catalyst generates a pentadienyl cation, which undergoes a thermally allowed 4π conrotatory electrocyclization as dictated by the Woodward-Hoffman rules. This generates an oxyallyl cation which undergoes an elimination reaction to lose a β-hydrogen. Subsequent tautomerization of the enolate produces the cyclopentenone product.
An electrocyclic reaction is the concerted interconversion of a conjugated polyene and a cycloalkene. Electrocyclic reactions are induced either thermally or photochemically.
In electrocyclic reactions either a ring is formed with the generation of a new σ bond and the loss of a π bond (i.e., gain of one σ bond and loss of one π bond) or ring is broken with the loss of one σ bond and gain of one π bond.
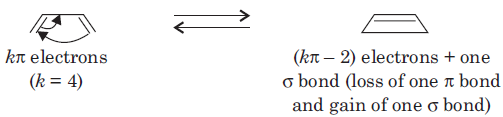
When performing an electrocyclic reaction, it is often desirable to predict the cis/trans geometry of the reaction's product. The first step in this process is to determine whether a reaction proceeds through conrotation or disrotation.
Information Booster
Trimethylsilyl and other silyl groups stabilize a positive charge on a β carbon and are lost very easily. They can be thought of as very reactive protons or ‘super protons’.