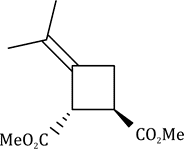
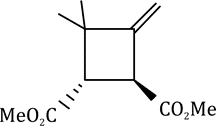
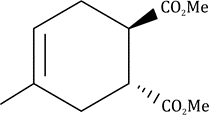
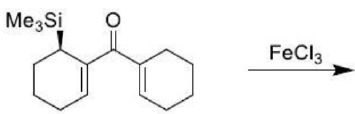
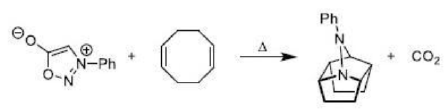
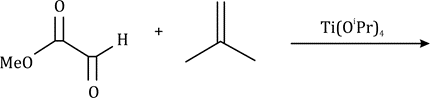
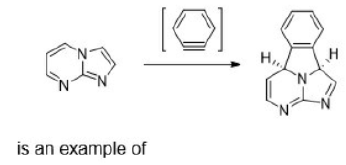
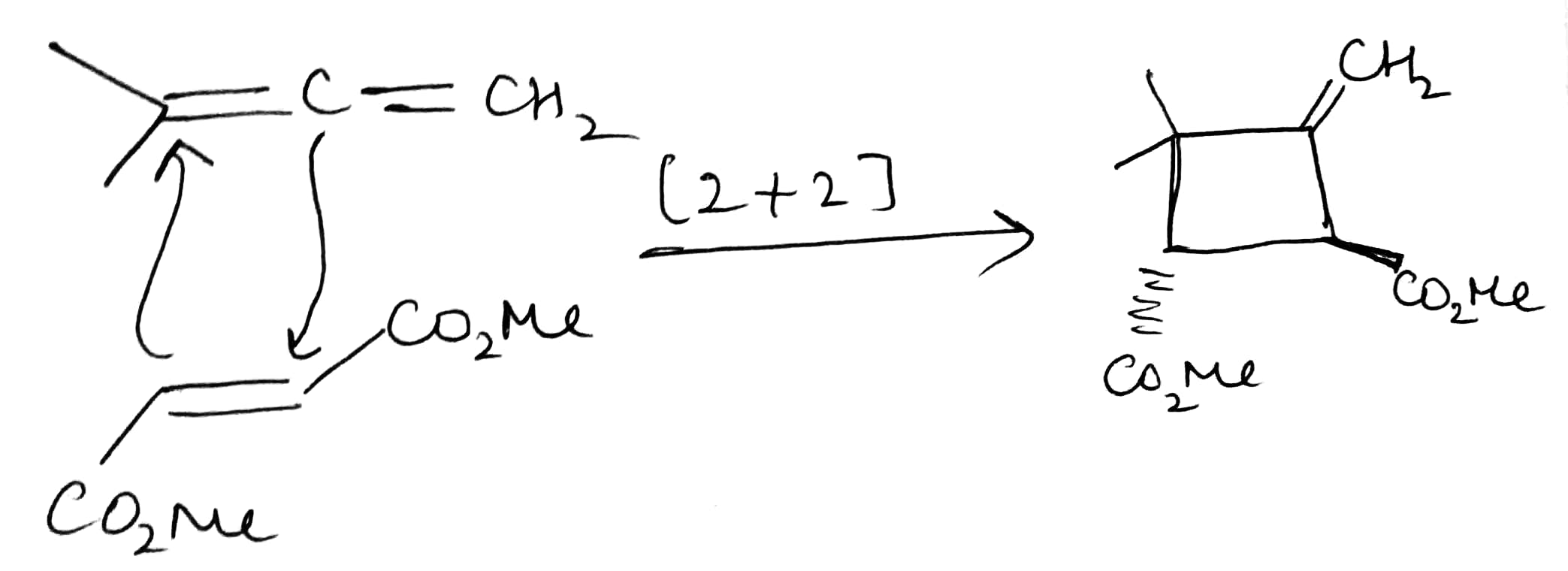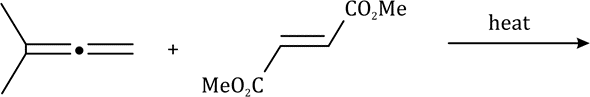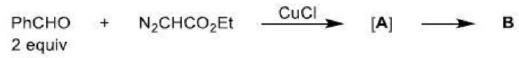Correct option is B
A cycloaddition is a reaction in which two unsaturated molecules undergo an addition reaction to yield a cyclic product. Formation of cyclic product takes place at the expense of one π (pi) bond in each of the reacting partner and gain of two σ (sigma) bonds at the end of the both
components having π ( pi ) bonds. Thus, in this reaction there is loss of two π (pi) bonds of the reactants and gain of two σ (sigma) bonds in the product.
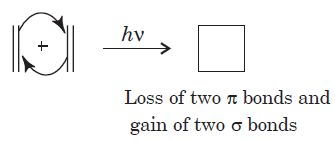
The above reaction is a [2 + 2] cycloaddition reaction because the reaction involves two electrons from one reacting component and also two electrons from the other.
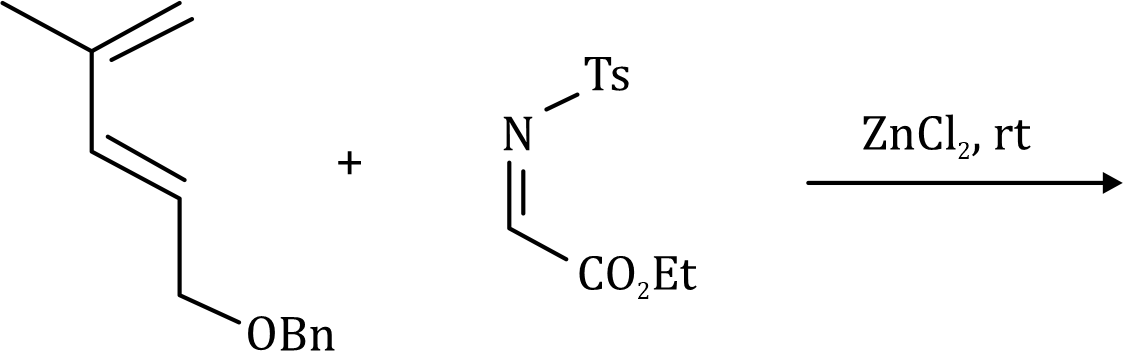
In the given question, the less hindered double bond of allene will participate in the reaction. The given alkene is a trans isomer and will remain so in the product.