Correct option is D
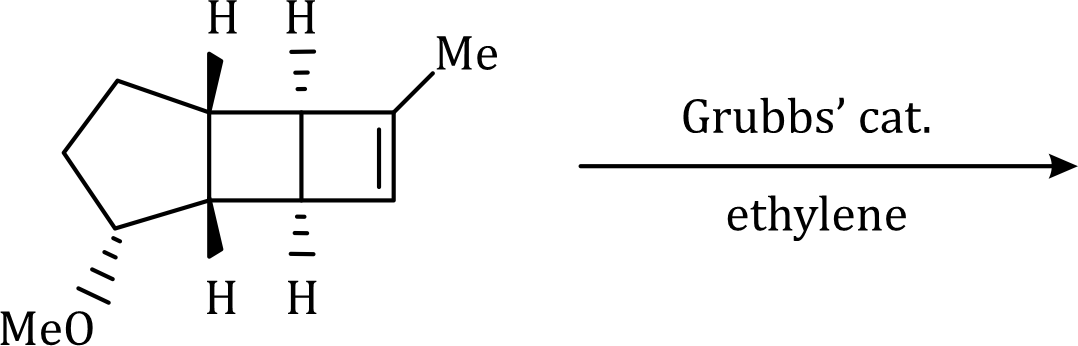
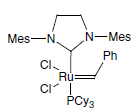
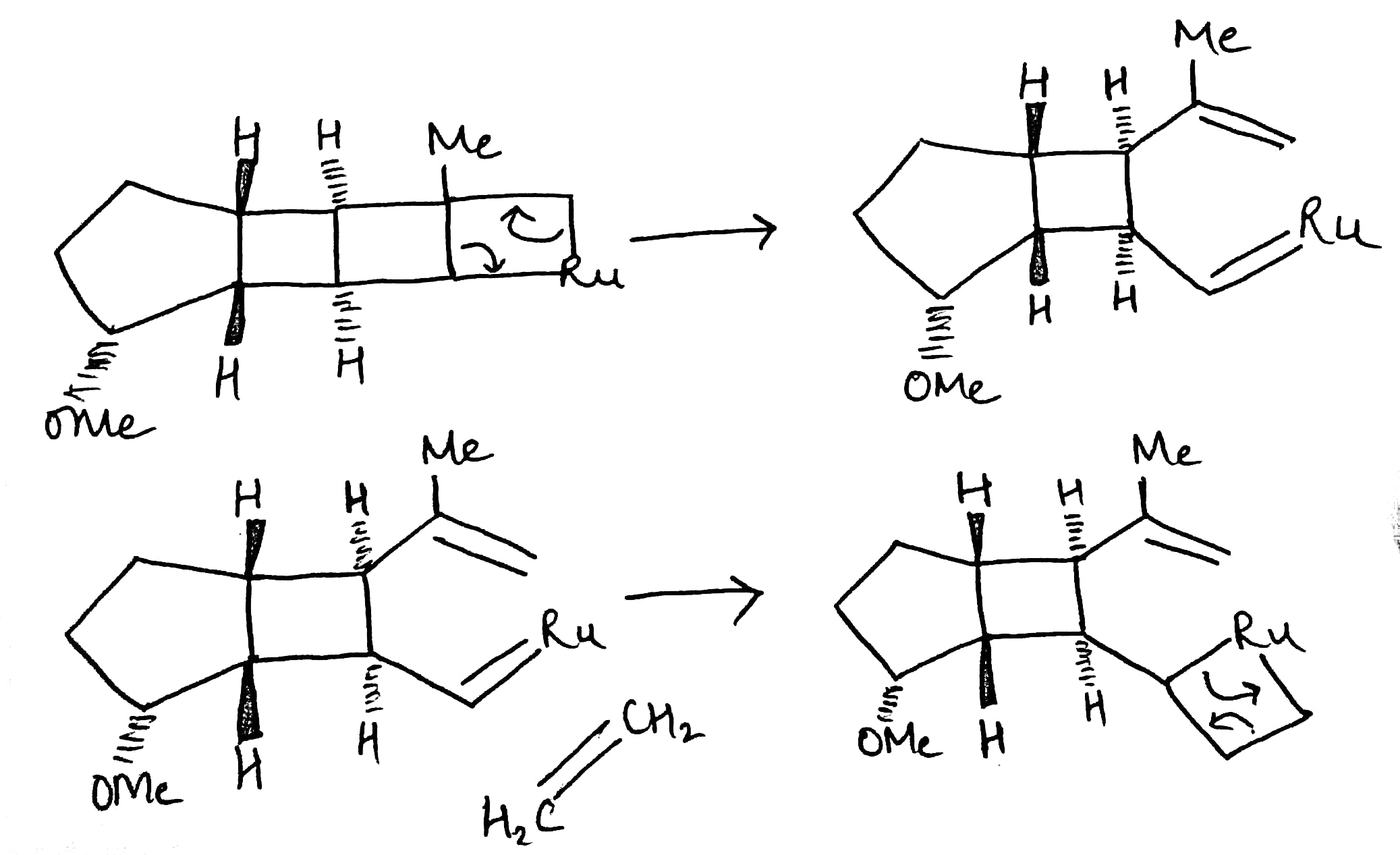
A significant development for the selective synthesis of alkenes makes use of alkene metathesis. Metathesis, as applied to two alkenes, refers to the transposition of the alkene carbon atoms, such that two new alkenes are formed . The reaction is catalysed by various transition-metal alkylidene (carbene) complexes, particularly those based on ruthenium or molybdenum. The ruthenium catalyst, developed by Grubbs, is the most popular, being more stable and more tolerant of many functional groups.
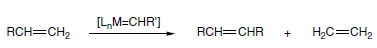
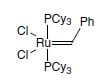
Ruthenium complexes which have resistance to oxygen and moisture as complex, have been found to be highly active metathesis catalysts.
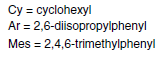
Alkene metathesis occurs by way of an intermediate metallacycle, followed by ring opening to give either the starting materials or one of the new alkenes and a new metallocarbene complex. Further metallocycle formation using another alkene and ring-opening provides the other product alkene and recovered catalyst to continue the cycle.
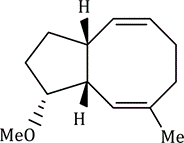
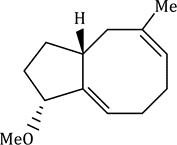
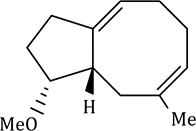
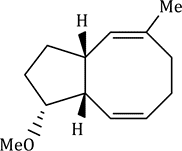
The reaction has shown considerable use in organic synthesis for ring formation (ring-closing metathesis, RCM). The method is not only effective for the preparation of five- and six-membered rings, but can be applied to medium and large ring formation. This has made it popular for the synthesis of many different substituted carbocyclic and heterocyclic ring systems.
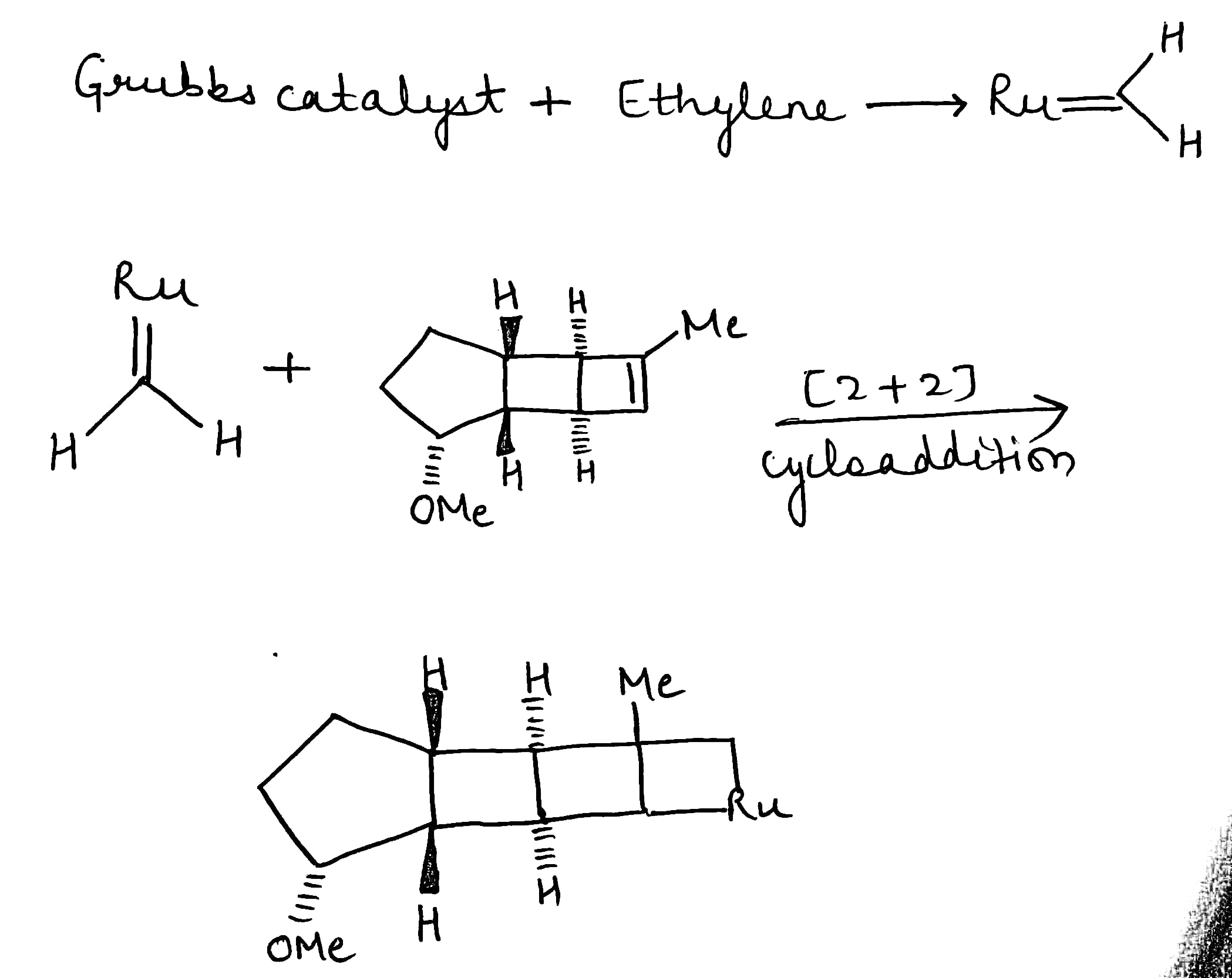
A cycloaddition is a reaction in which two unsaturated molecules undergo an addition reaction to yield a cyclic product. Formation of cyclic product takes place at the expense of one π (pi) bond in each of the reacting partner and gain of two σ (sigma) bonds at the end of the both components having π ( pi ) bonds. Thus, in this reaction there is loss of two π (pi) bonds of the reactants and gain of two σ (sigma) bonds in the product.
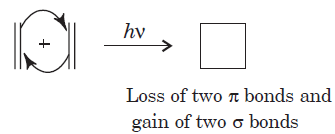
The reaction in the above equation is a [2 + 2] cycloaddition reaction because the reaction involves two electrons from one reacting component and also two electrons from the other.
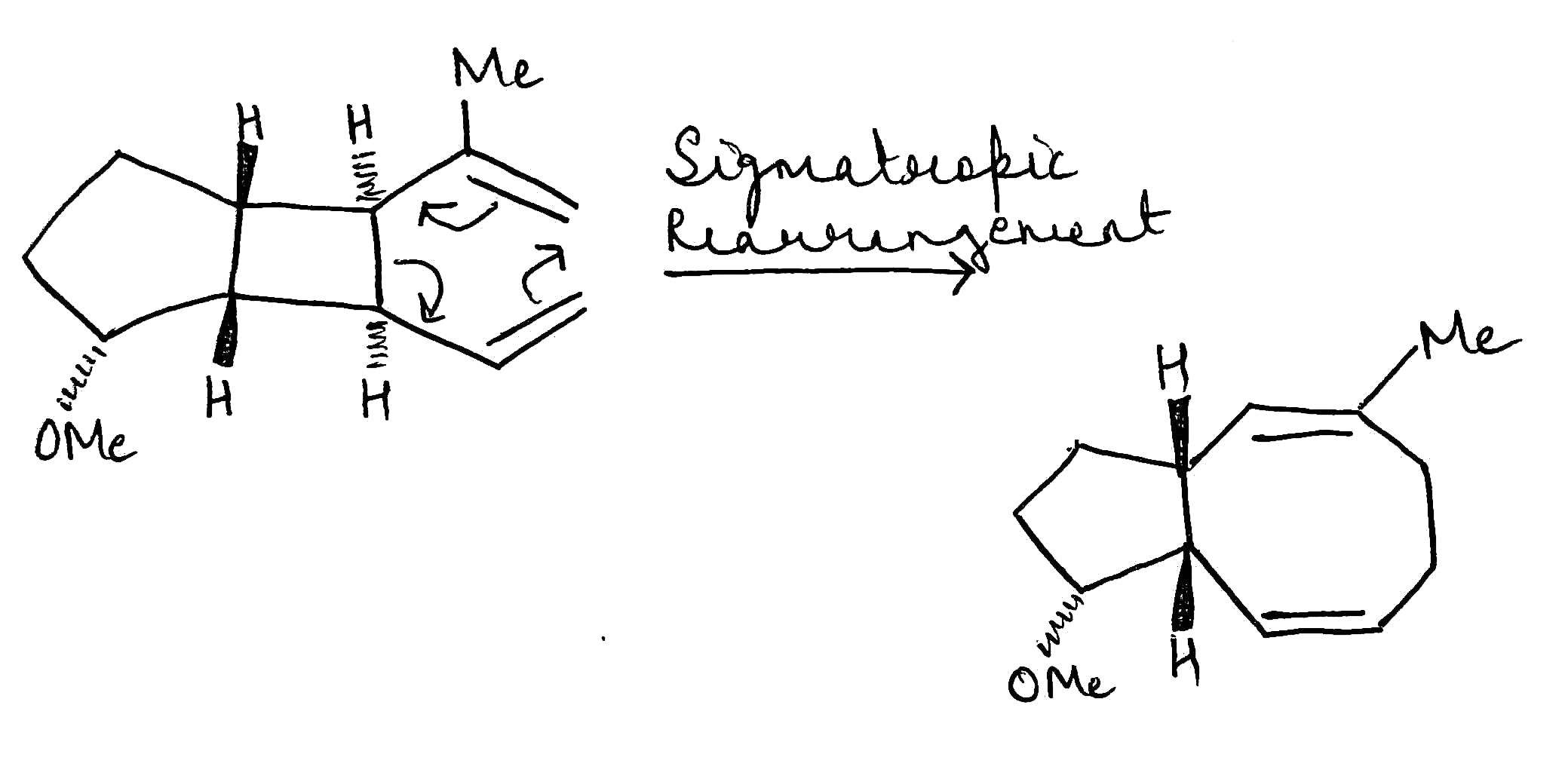
The [3,3]-sigmatropic rearrangement has been used most in organic synthesis. The reaction has found particular use for the stereocontrolled preparation of carbon–carbon bonds. In the course of the reaction both a new carbon–carbon single bond and a new carbon–carbon double bond are formed. The high levels of stereoselectivity arise as a result of a highly ordered six-membered-ring transition state, which (unless constrained conformationally) prefers a chair shape. The reaction involves the interconversion of 1,5-dienes and the all-carbon system is known as the Cope rearrangement.