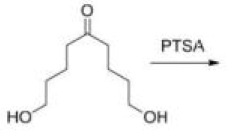
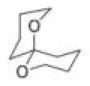
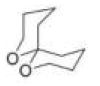
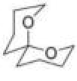
Correct option is C
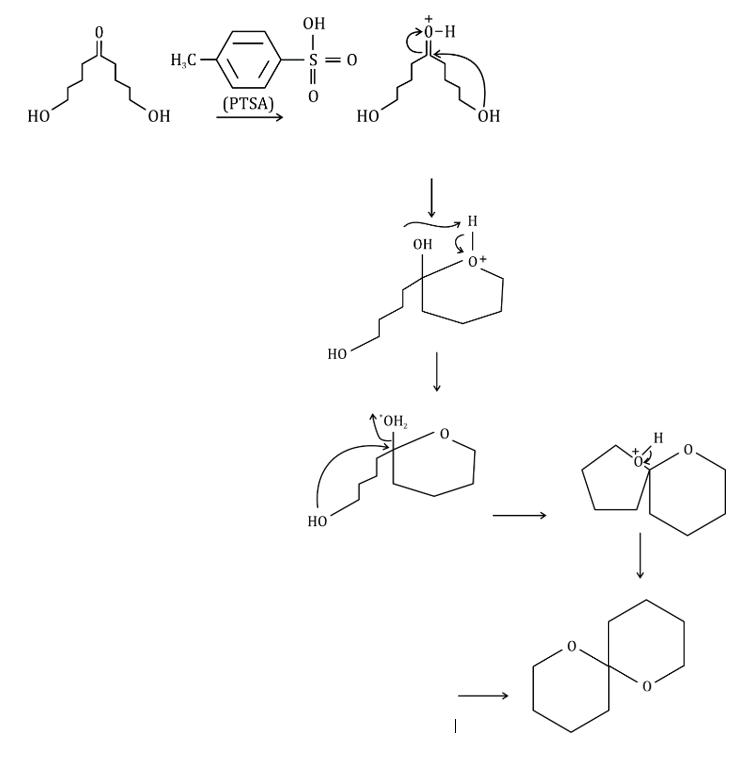
Consider the orientation of each C–O bond with respect to the ring that it is not part of: it is possible to have each C–O axial or equatorial, and there are three possible arrangements (three conformations).
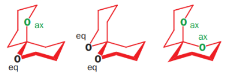
According to NMR, this compound exists entirely in one conformation: in which each oxygen is axial on the other ring. Only in this conformation can both C-O bonds benefit from the anomeric effect-this is often known as the double anomeric effect. In organic chemistry, the anomeric effect is a stereoelectronic effect that describes the tendency of heteroatomic substituents adjacent to a heteroatom within a cyclohexane ring to prefer the axial orientation instead of the less-hindered equatorial orientation that would be expected from steric considerations.