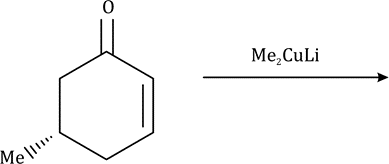
Correct option is B
Conjugate addition is an important C-C bond formation strategy available to the organic chemist. Organometallic reagents may add in a 1,2- or 1,4-manner to α,β-unsaturated carbonyl compounds.
The organocopper reagents used for conjugate additions to enones are homocuprates, heterocuprates, higher-order cuprates, and Grignard reagents in the presence of catalytic amounts of copper salts (CuX). Addition of organocopper reagents to α,β-unsaturated carbonyl compounds (enones and conjugated esters) generates enolates with concomitant introduction of an organic group at the β-position.
Conjugate addition of lithiocuprates to α,β-unsaturated ketones and esters may proceed via a initial reversible copper(I)-olefin-lithium association, which then undergoes oxidative addition followed by reductive elimination.
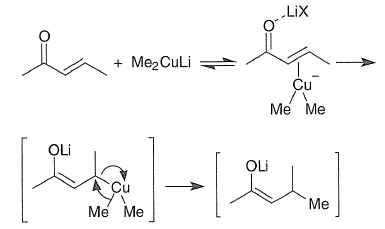
Stereochemistry of 1,4 addition reactions
Factors controlling the stereochemistry of conjugate additions are not well understood. Mixtures of isomers are often produced, but generally one isomer predominates. Both steric and electronic factors play a role. Generally, Michael-type additions have late and hence product like-and chair like-transition states. In the example shown below, for stereoelectronic reasons antiparallel attack by the nucleophilic "CH3" is favored over parallel attack.
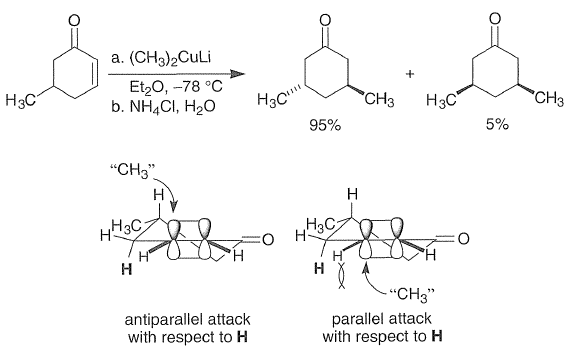
As there is no plane of symmetry in the trans product (95%), hence it is chiral.