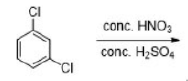
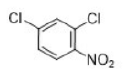
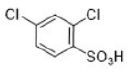
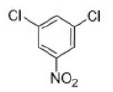
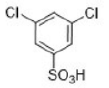
Correct option is A
Nitration of benzene
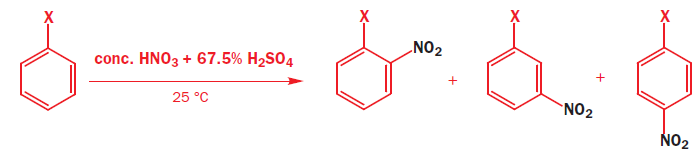
Aromatic nitration requires very powerful reagents, the most typical being a mixture of concentrated nitric and sulfuric acids.
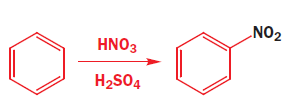
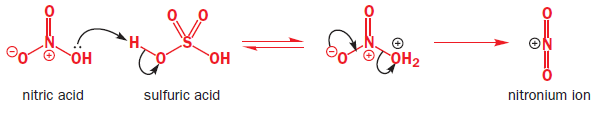
The first steps are the formation of a very powerful electrophile, none other than NO2+ , by the interaction of the two strong acids. Sulfuric acid is the stronger and it protonates the nitric acid on the OH group so that a molecule of water can leave.
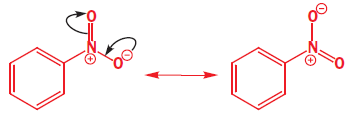
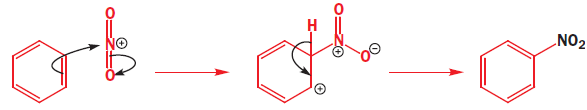
The nitronium ion ( NO2+) is linear with an sp hybridized nitrogen at the centre. Benzene attacks the positively charged nitrogen atom but one of the N=O bonds must be broken at the same time to avoid five-valent nitrogen.
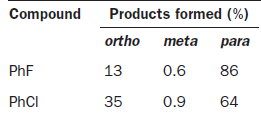
The halogens deactivate the ring towards electrophilic attack but direct ortho and para. The only way this makes sense is if there are two opposing effects—electron donation by conjugation and electron withdrawal by induction. The halogen has three lone pairs, one of which may conjugate with the ring. However, there are two mismatching aspects to this conjugation: lone pair orbital size and electronegativity.
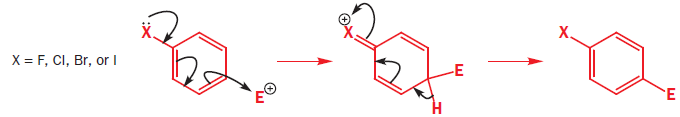
When Cl, Br, or I is the substituent, there is a size mismatch, and therefore a poor overlap, between the 2p orbitals from the carbon atoms and the p orbitals from the halogen (3p for chlorine, 4p for bromine, and 5p for iodine). The more electronegative a substituent, the better it is at withdrawing electrons by induction.
Most electron density is removed first from the ortho positions by induction, then from the meta positions, and then from the para position. Any conjugation of the lone pairs on chlorine with the pi system would increase the electron density in the ortho and para positions. Both effects favour the para position and this is where most substitution occurs. Data for the rate and the products of nitration of halobenzenes show these opposing effects clearly.