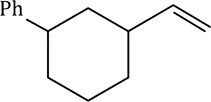
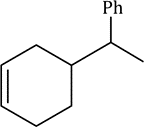
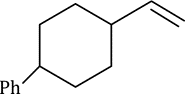
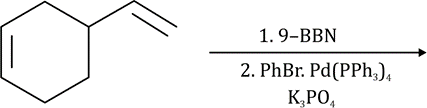Correct option is A
Hydroboration
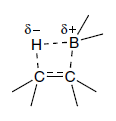
Organoboranes are obtained by addition of borane or alkyl boranes to alkenes (or alkynes). Borane itself can be prepared by reaction of boron trifluoride etherate with sodium borohydride. Borane exists as a dimer, but solutions containing an electron donor, such as an ether, amine or sulfide, allow adduct formation.
The most important synthetic application of borane is for the preparation of alkyl boranes by addition to alkenes, a process known as hydroboration. Borane and its derivatives can also be used for reduction. The hydroboration reaction has been applied to a large number of alkenes of widely differing structures. In nearly all cases the addition proceeds rapidly at room temperature, and only the most hindered alkenes do not react.
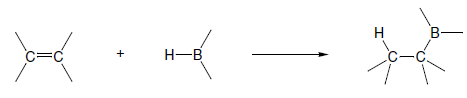
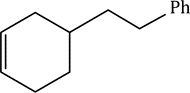
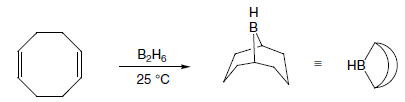
Hydroboration of mono- and disubstituted alkenes with borane gives rise typically to a trialkylborane product. However, trisubstituted alkenes normally give a dialkylborane and tetrasubstituted alkenes form only the monoalkylboranes. The extent of hydroboration may also be controlled by the stoichiometry of alkene and borane. This has been exploited in the preparation of a number of mono and dialkylboranes that are less reactive and more selective than borane itself. Important in this respect is 9-BBN (9-borabicyclo[3.3.1]nonane), formed by addition of borane to 2-methyl-2-butene, 2,3-dimethyl-2-butene and 1,5-cyclo-octadiene respectively. These partially alkylated boranes may themselves be used to hydroborate less-hindered alkenes.
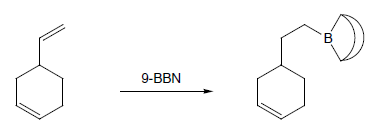
9-BBN is more sensitive to the structure of the alkene than is borane itself. Terminal alkenes react more rapidly than internal alkenes and Z-alkenes more rapidly than E-alkenes. This sometimes allows the selective hydroboration of one double bond in a diene or triene. Thus, the non-conjugated diene is readily hydroborated to give the organoborane, with no hydroboration of the more-hindered alkene.
Suzuki coupling
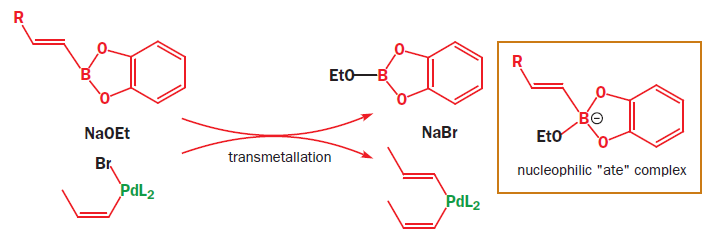
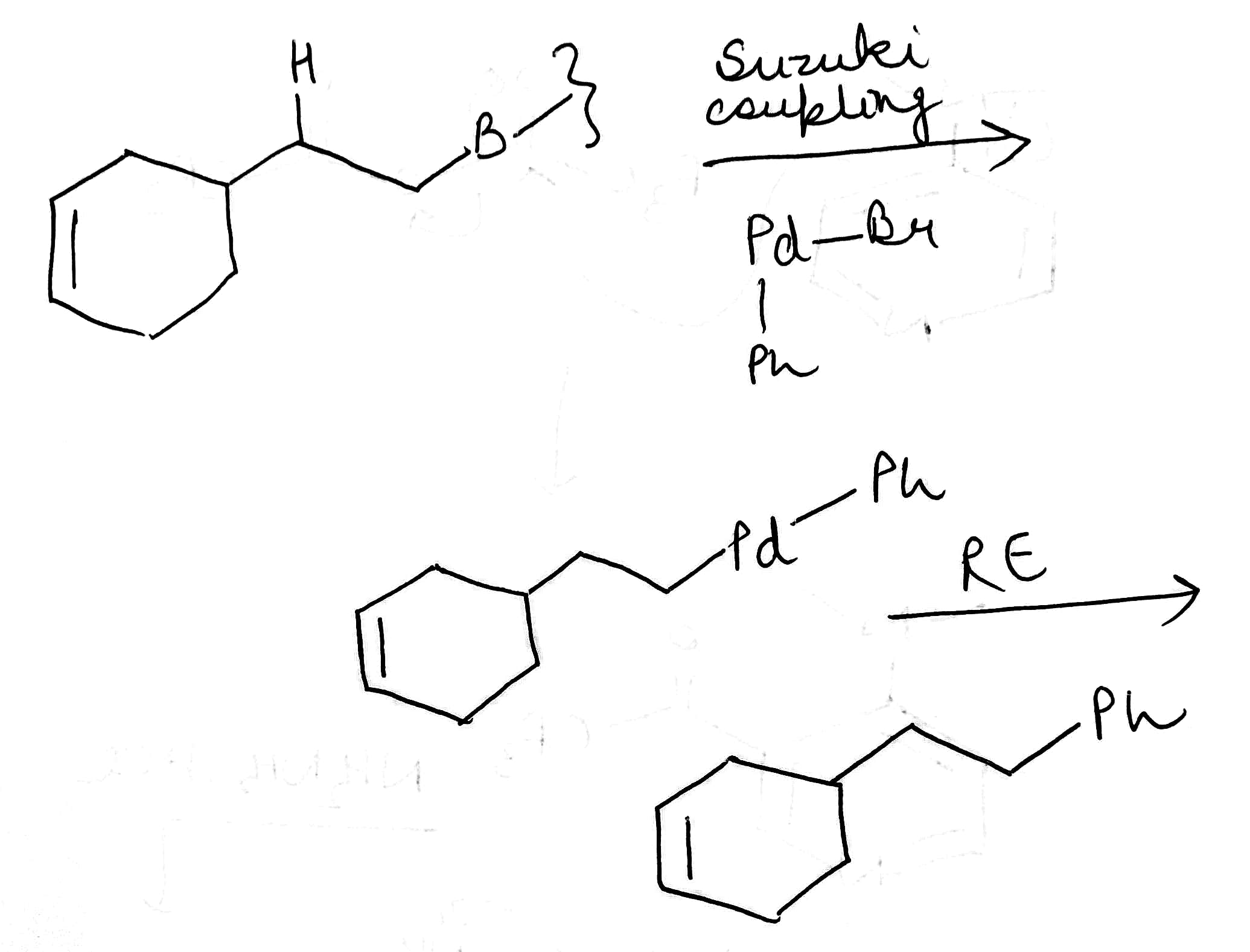
The Suzuki coupling of a boronic acid with a halide or triflate has developed into one of the most important cross-coupling reactions, totalling about a quarter of all current palladium-catalysed cross-coupling reactions. The hydroboration is generally regioselective for the less hindered position and addition of boron and hydrogen occurs cis stereospecifically.
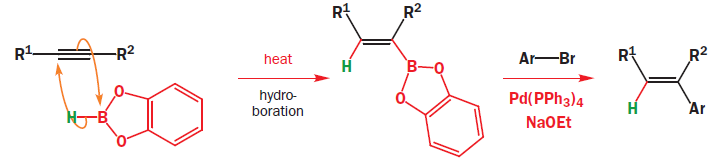
Oxidative addition of the vinylic or aromatic halide to the palladium(0) complex generates a palladium(II) intermediate. This then undergoes a transmetallation with the alkenyl boronate, from which the product is expelled by reductive elimination, regenerating the palladium(0) catalyst. The important difference is the transmetallation step, which explains the need for an additional base, usually sodium or potassium ethoxide or hydroxide, in the Suzuki coupling. The base accelerates the transmetallation step leading to the borate directly presumably via a more nucleophilic ‘ate’ complex.