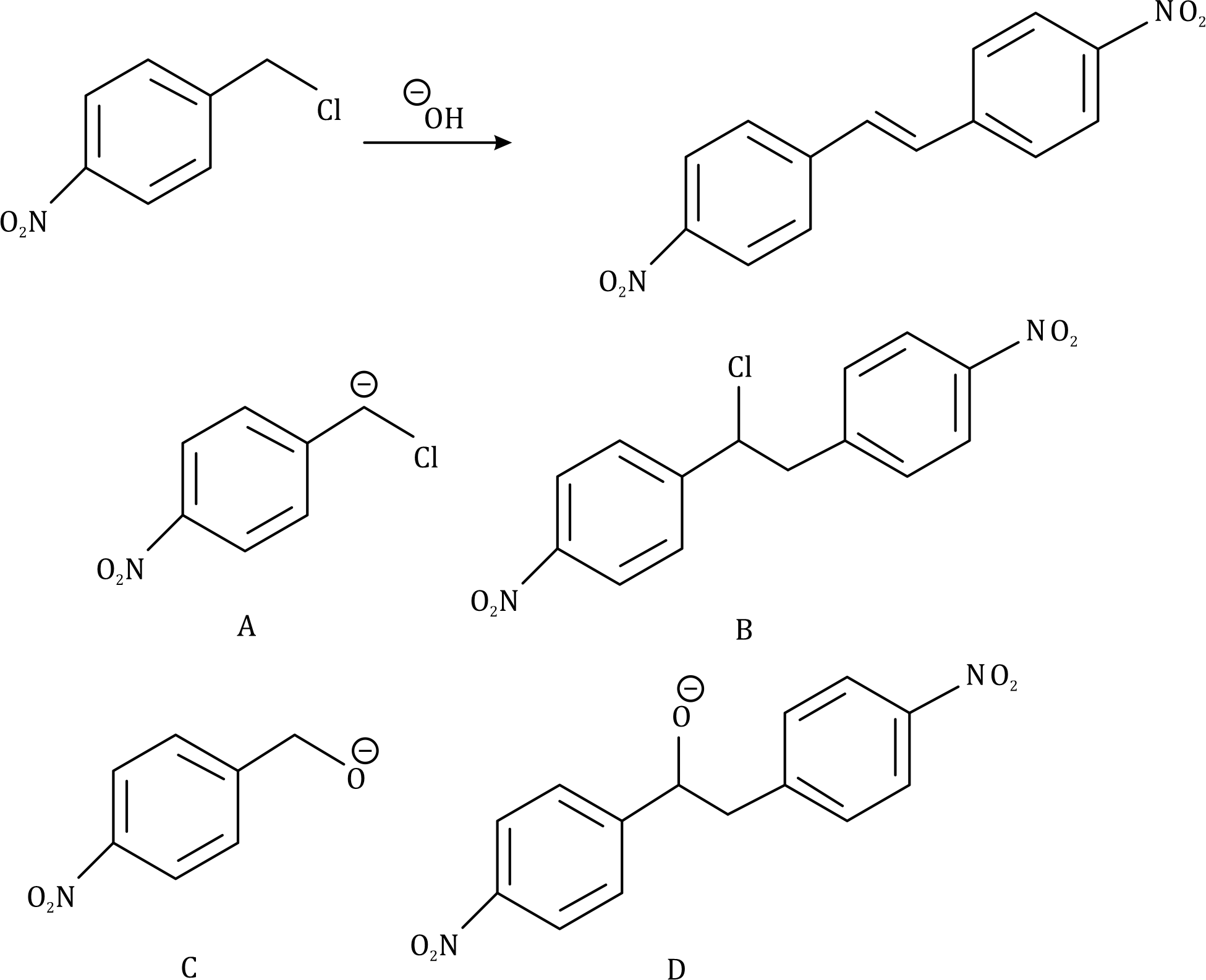
Correct option is B
Hydroxide ion (-OH) will act as a base and abstract the acidic hydrogen (H+). The carbanion generated will get stabilized by the electron withdrawing NO2 group (present at para position) and the Cl atom. The generated carbanion will attack another molecule of the substrate.
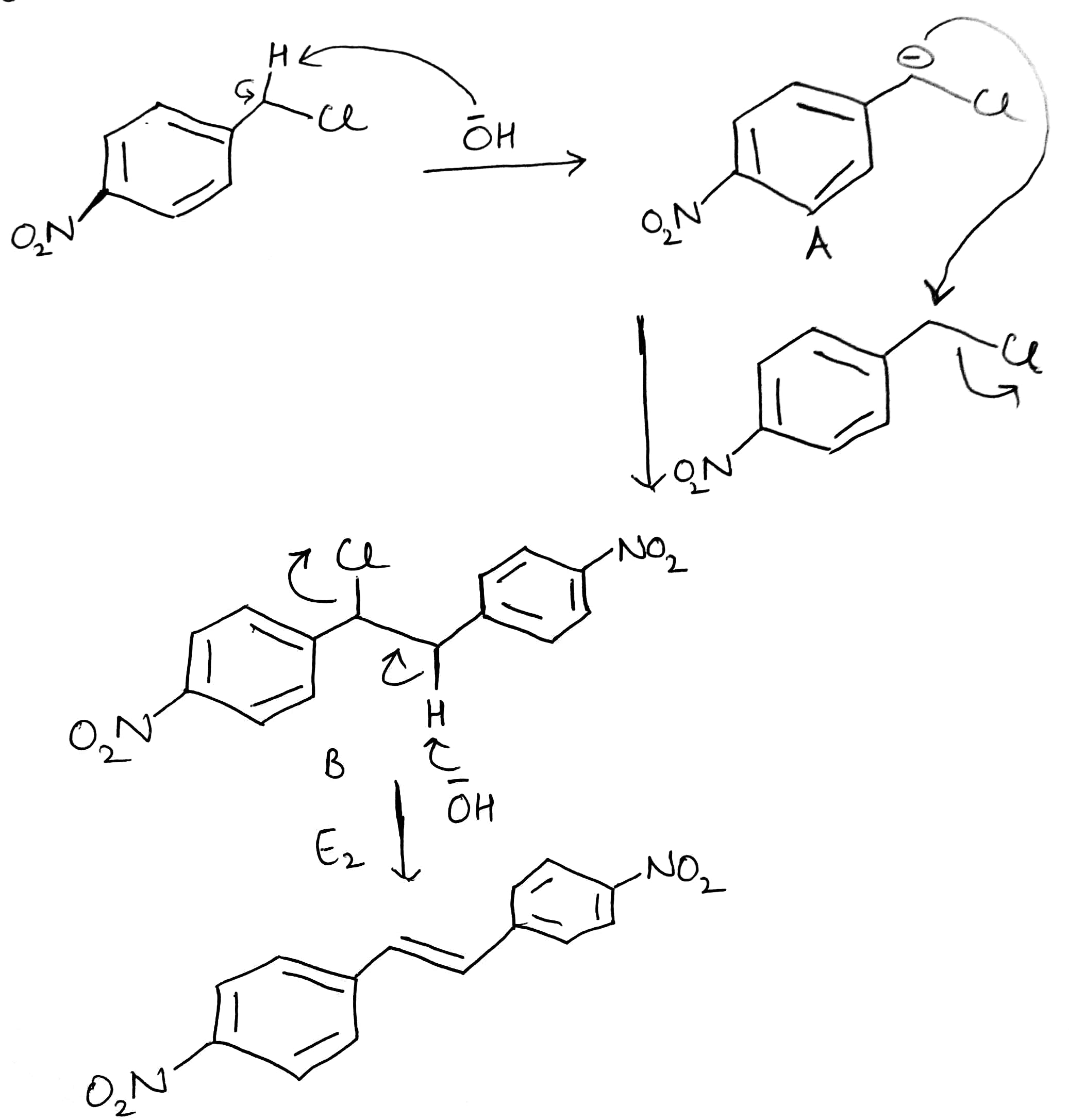
E2 elimination
An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one- or two-step mechanism. The one-step mechanism is known as the E2 reaction, and the two-step mechanism is known as the E1 reaction.
The E2 mechanism, where E2 stands for bimolecular elimination, involves a one-step mechanism in which carbon-hydrogen and carbon-halogen bonds break to form a double bond (C=C Pi bond).The specifics of the reaction are as follows:
1.E2 is a single step elimination, with a single transition state.
2.It is typically undergone by primary substituted alkyl halides, but is possible with some secondary alkyl halides and other compounds.
3.The reaction rate is second order, because it's influenced by both the alkyl halide and the base (bimolecular).
4.Because the E2 mechanism results in the formation of a pi bond, the two leaving groups (often a hydrogen and a halogen) need to be antiperiplanar. Antiperiplanar, describes the A−B−C−D bond angle in a molecule. In this conformer, the dihedral angle of the A−B bond and the C−D bond is greater than +150° or less than −150°.
5.E2 typically uses a strong base. It must be strong enough to remove a weakly acidic hydrogen.
6.In order for the pi bond to be created, the hybridization of carbons needs to be lowered from sp3 to sp2.