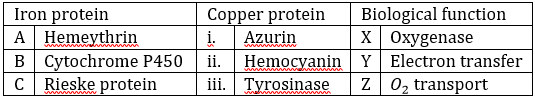
Correct option is B
Nitrite reductase refers to any of several classes of enzymes that catalyze the reduction of nitrite. There are two classes of NIR's. A multi heme enzyme reduces NO2- to a variety of products. Copper containing enzymes carry out a single electron transfer to produce nitric oxide.
Copper Nitrite Reductases are found in many different fungi and bacteria. What is common to all CuNIR is the presence of at least one type 1 copper center in the protein.
Type I copper centres (T1Cu) are characterized by a single copper atom coordinated by two histidine residues and a cysteine residue in a trigonal planar structure, and a variable axial ligand. In class I T1Cu proteins (e.g. amicyanin, plastocyanin and pseudoazurin) the axial ligand is the sulfur of methionine, whereas amino acids other than methionine (e.g. glutamine) give rise to class II T1Cu copper proteins. Azurins contain the third type of T1Cu centres: besides a methionine in one axial position, they contain a second axial ligand (a carbonyl group of a glycine residue). T1Cu-containing proteins are usually called "cupredoxins", and show similar three-dimensional structures, relatively high reduction potentials (> 250 mV), and strong absorption near 600 nm (due to S→Cu charge transfer), which usually gives rise to a blue colour. Cupredoxins are therefore often called "blue copper proteins". This may be misleading, since some T1Cu centres also absorb around 460 nm and are therefore green.
Each type 1 Cu is strongly bonded to a thiolate sulfur from a cysteine, two imidazole nitrogens from different Histidine residues, and a sulfur atom of an axial Methionine ligand. This induces a distorted tetrahedral molecular geometry.
The cysteine ligated to the type 1 Cu center is located directly next to a Histidine in the primary structure of the amino acids. This Histidine is bound to the Type 2 Cu center responsible for binding and reducing nitrite. This Cys-His bridge plays an important role in facilitating rapid electron transfer from the type 1 center to the type 2.
Type II copper centres (T2Cu) exhibit a square planar coordination by N or N/O ligands. Since no sulfur ligation is present, the optical spectra of these centres lack distinctive features. T2Cu centres occur in enzymes, where they assist in oxidations or oxygenations.
Proposed mechanism
The type 2 copper center of a copper nitrite reductase is the active site of the enzyme. The Cu is bound by nitrogens of two Histidines from one monomer, and bound by one Histidine from another monomer; the Cys-His bridge to the type 1 Cu. This gives the molecule a distorted tetrahedral geometry. In the resting state, the Cu is also binding a water molecule that is displaced by nitrite.
As nitrite displaces water, Cu is bound by both oxygens in a bidentate fashion. A nearby Aspartic acid residue hydrogen bonds to one of the newly formed oxygen ligands. An incoming electron reduces the Cu from oxidation state (II) to (I). This change facilitates a shift in nitrite binding so that the nitrogen is bound to Cu, and one oxygen has an extended bond length due to hydrogen bonding. A second hydrogen bond forms from Histidine or a nearby water molecule and leads to the cleavage of the N-O bond. The Cu is now five coordinate bonded to nitric oxide and water. Nitric oxide is released as Cu is oxidized to state (II) and returns to the resting configuration.
Additional Knowledge
Type III copper centres (T3Cu) consist of a pair of copper centres, each coordinated by three histidine residues. These centres are present in some oxidases and oxygen-transporting proteins (e.g. hemocyanin and tyrosinase).