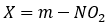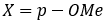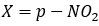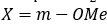Correct option is C
The addition-elimination mechanism involves addition of the nucleophile followed by elimination of the leaving group. The nucleophilic aromatic substitution reaction proceeds via the addition-elimination mechanism. A typical nucleophilic aromatic substitution has:
a. An oxygen, nitrogen, or cyanide nucleophile
b. A halide for a leaving group
c. A carbonyl, nitro, or cyanide group ortho or para to the leaving group
Information Booster
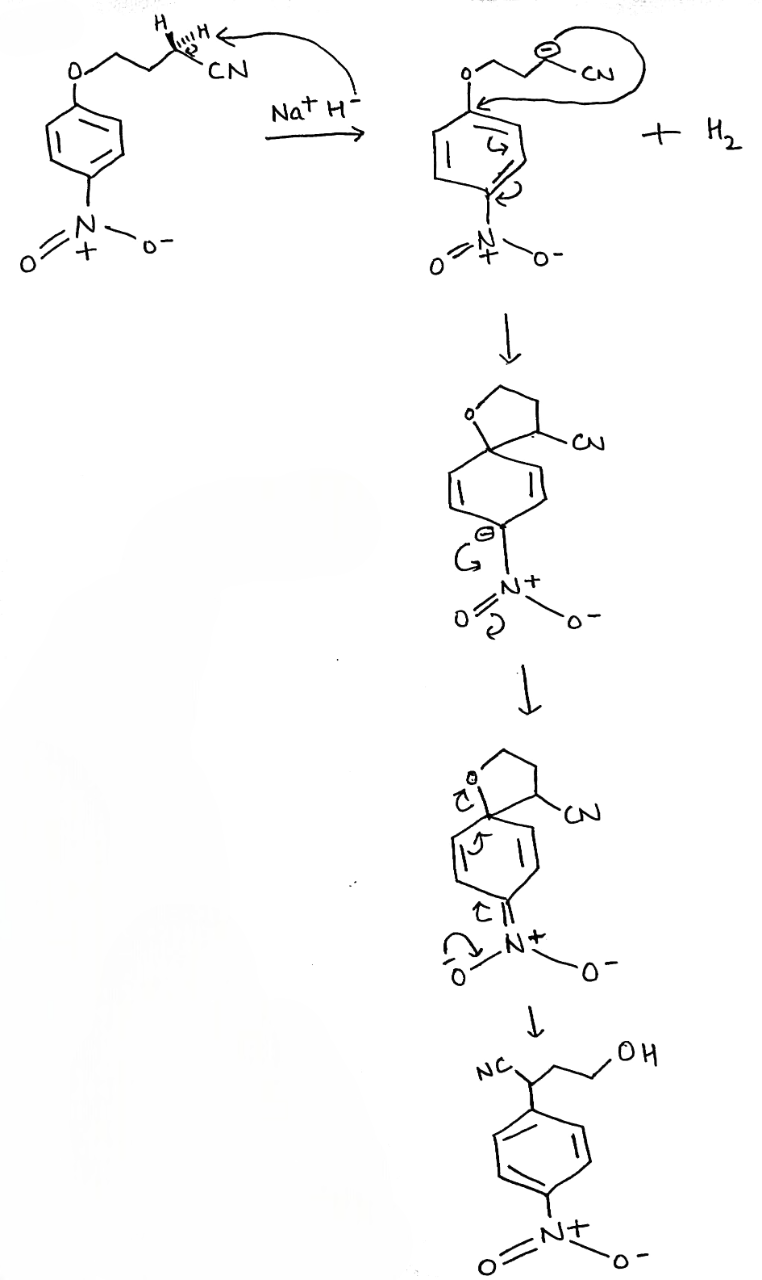
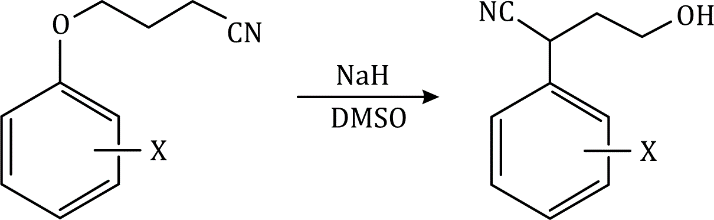
It is not necessary to have a carbonyl group-any electron-withdrawing group will do-the only requirement is that the electrons must be able to get out of the ring into this anion-stabilizing group. Here is an example with a para-nitro group.
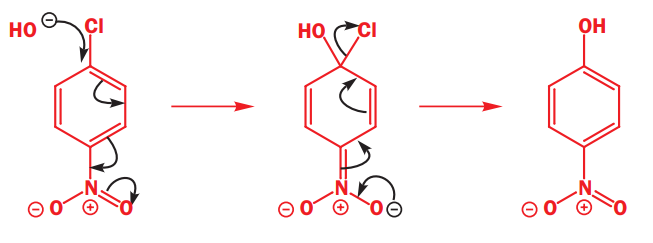
In a two-step mechanism, one step is slower and rate-determining; the other is unimportant to the rate. In the mechanism for nucleophilic aromatic substitution, it is the first step that is slower because it disturbs the aromaticity. The second step restores the aromaticity and is faster.
The nucleophile is a good one, the negative charge can be pushed through on to the oxygen atom(s) of the nitro group.
Any anion-stabilizing (electron-withdrawing) group ortho or para to a potential leaving group can be used to make nucleophilic aromatic substitution possible.