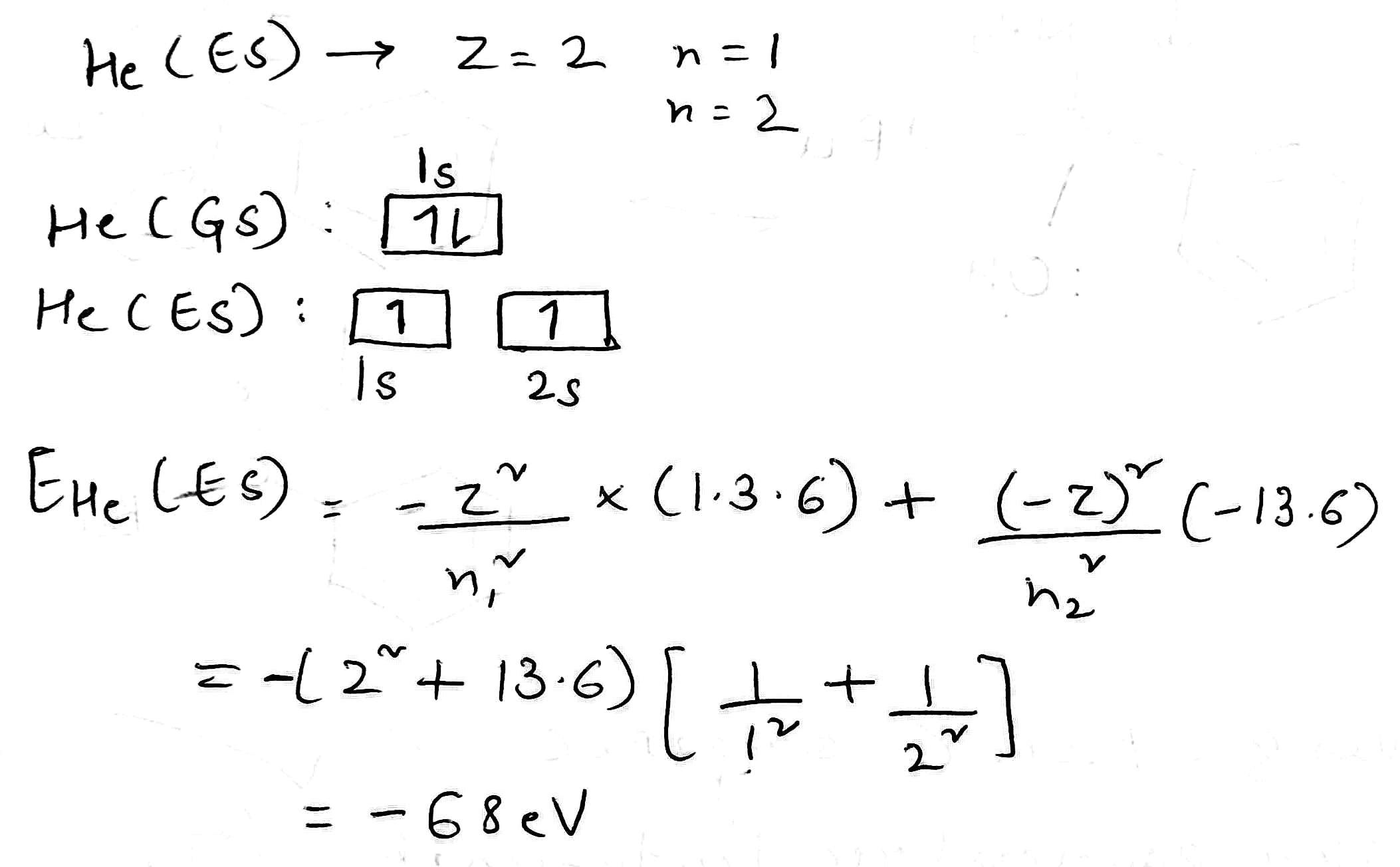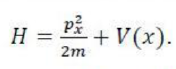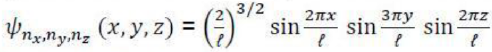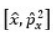Correct option is A
The Bohr model or Rutherford–Bohr model was the first successful model of the atom. It consists of a small, dense nucleus surrounded by orbiting electrons. It is analogous to the structure of the Solar System, but with attraction provided by electrostatic force rather than gravity, and with the electron energies quantized (assuming only discrete values). The model's key success lies in explaining the Rydberg formula for hydrogen's spectral emission lines.
In classical mechanics, if an electron is orbiting around an atom with period T, and if its coupling to the electromagnetic field is weak, so that the orbit doesn't decay very much in one cycle, it will emit electromagnetic radiation in a pattern repeating at every period, so that the Fourier transform of the pattern will only have frequencies which are multiples of 1/T.
However, in quantum mechanics, the quantization of angular momentum leads to discrete energy levels of the orbits, and the emitted frequencies are quantized according to the energy differences between these levels. This discrete nature of energy levels introduces a fundamental departure from the classical radiation law, giving rise to distinct spectral lines in the emitted radiation.
Bohr assumes that the electron is circling the nucleus in an elliptical orbit obeying the rules of classical mechanics, but with no loss of radiation due to the Larmor formula.
Denoting the total energy as E, the negative electron charge as e, the positive nucleus charge as K=Z|e|, the electron mass as me, half the major axis of the ellipse as a, he starts with these equations:
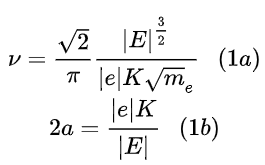
E is assumed to be negative, because a positive energy is required to unbind the electron from the nucleus and put it at rest at an infinite distance.
Eq. (1a) is obtained from equating the centripetal force to the Coulombian force acting between the nucleus and the electron, considering that
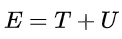
(where T is the average kinetic energy and U the average electrostatic potential), and that for Kepler's second law, the average separation between the electron and the nucleus is a.
Eq. (1b) is obtained from the same premises of eq. (1a) plus the virial theorem, stating that, for an elliptical orbit,
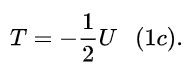
Then Bohr assumes that
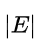
is an integer multiple of the energy of a quantum of light with half the frequency of the electron's revolution frequency,

He further assumes that the orbit is circular, i.e. a=r , and, denoting the angular momentum of the electron as L, introduces the equation:
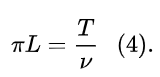
Eq. (4) stems from the virial theorem, and from the classical mechanics relationships between the angular momentum, the kinetic energy and the frequency of revolution.

An electron in the lowest energy level of hydrogen (n = 1) therefore has about 13.6 eV less energy than a motionless electron infinitely far from the nucleus. The next energy level (n = 2) is −3.4 eV. The third (n = 3) is −1.51 eV, and so on.