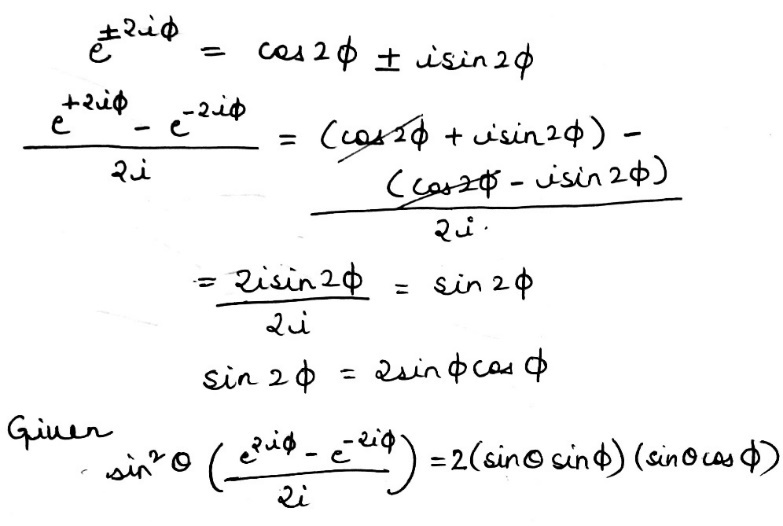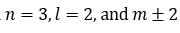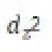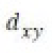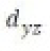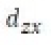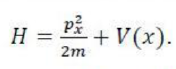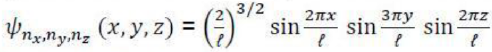Correct option is B
Quantum mechanical treatment of the hydrogen-like species

To describe the quantum mechanical state of the hydrogen-like species, we need a function ψ which depends on the six independent variables, viz., three coordinates of the nucleus and three for the electron.
If the function ψ is written as
Ψ=ψMψe
where ψM depends on the coordinates of the centre of mass and ψe on the internal coordinates, the Schrödinger equation separates into two independent equations; one for the motion of the atom as a whole and the other involving the internal coordinates of the atom. The latter one is of interest to us as it would give us the wave functions of the electron and the associated energies.
The Schrödinger equation involving the internal coordinates is then transformed into the spherical polar coordinates.
The Schrödinger equation splits into three equations if we write ψe as
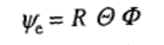
The function R involves only r,

involves only the angle θ and
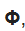
involves only angle
The three resultant equations can be solved for the functions R,

and
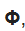
by suitable transformations. In order that these functions are finite, well- behaved and consistent with the boundary conditions, certain quantum restrictions are introduced. These restrictions appear in the form of quantum numbers.
The functions R depend only on r, therefore, they describe the distribution of the electron as a function of r from the nucleus. These functions depend upon two quantum numbers, n and l. The permitted values of these numbers are:
Principal quantum number: n = 1, 2, 3, 4, ...
Azimuthal quantum number: l = 0, 1, 2, ..., (n - 1)
We have different R functions for different values of n and l.
The functions

depend only on angle θ, therefore, they describe the distribution as a function of angle θ. These functions again, depend upon two quantum numbers, viz., l and m. Though the permitted values of m are 0, ± 1, ± 2, ..., ±l, the

functions depend only on the magnitude of m.
The functions
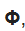
depend only on angle
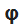
, therefore, they describe the distribution as a function of angle
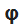
. These functions depend only on the value of m.
The two functions

and
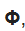
taken together give the angular distribution of the electron. Thus, the total wave function ψe which constitutes what is known as an orbital, depends on the quantum numbers n, l and m, i.e, we have different ψe functions (or orbitals) for different values of n, l and m and hence different behaviour of the electron in the atom.