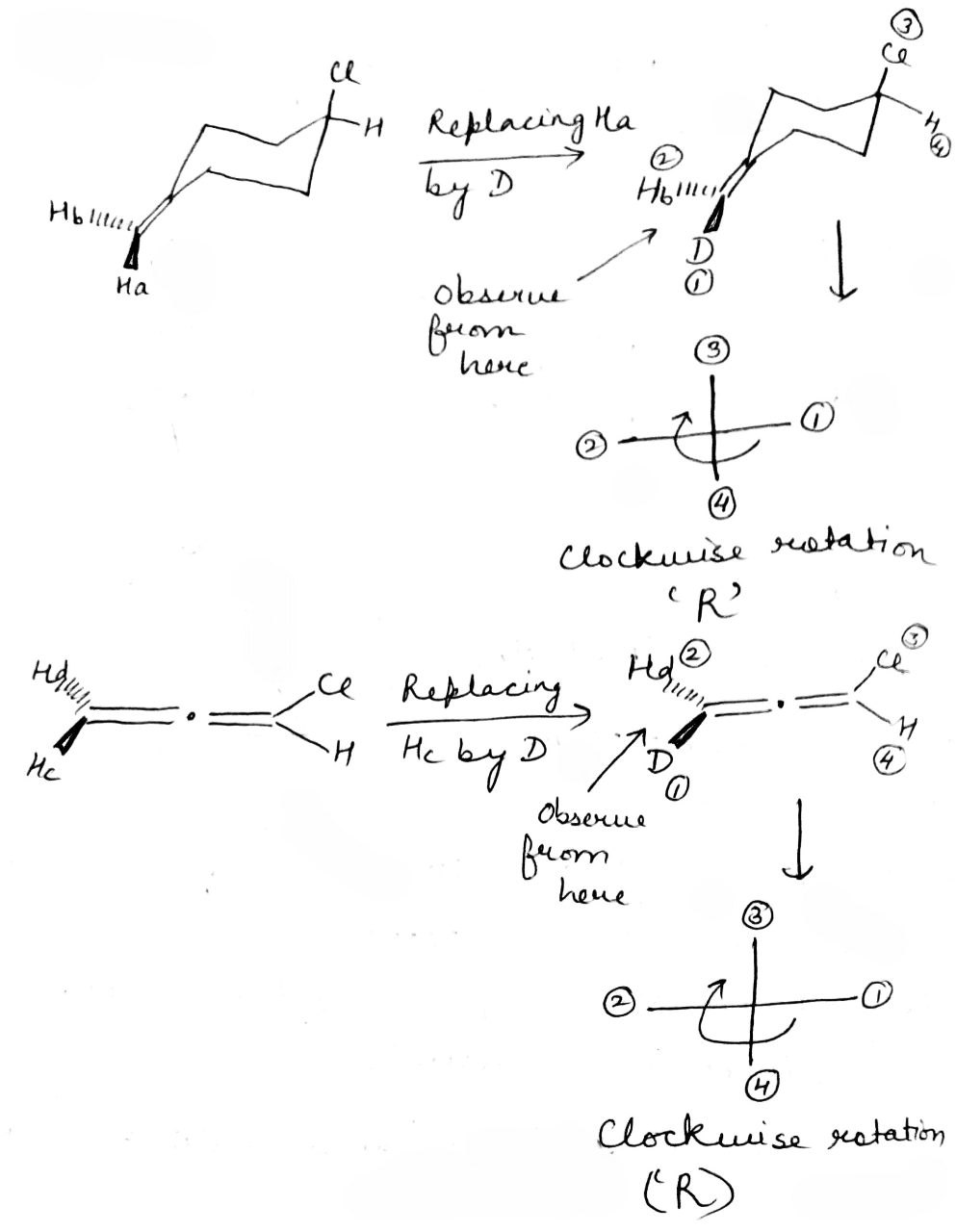
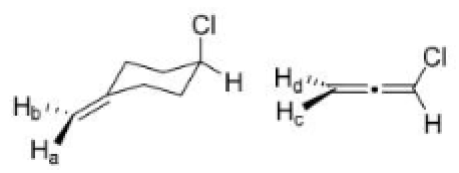
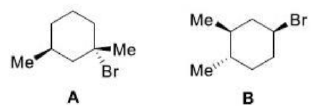
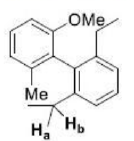
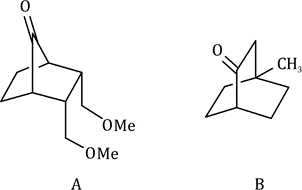
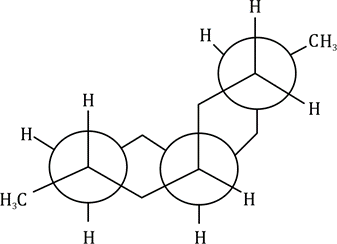
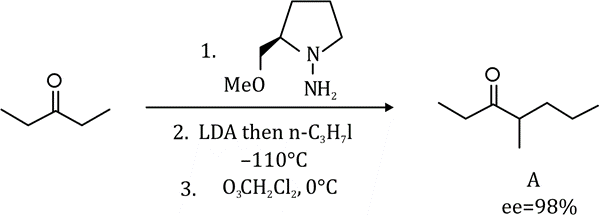
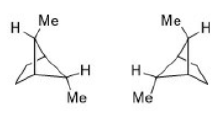
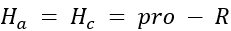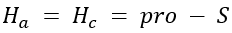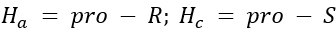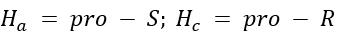Correct option is A
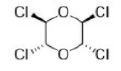
In chemistry, absolute configuration refers to the spatial arrangement of atoms within a molecular entity (or group) that is chiral, and its resultant stereochemical description. Absolute configuration is typically relevant in organic molecules where carbon is bonded to four different substituents. This type of construction creates two possible enantiomers.
Absolute configuration uses a set of rules to describe the relative positions of each bond around the chiral center atom. When the center is oriented so that the lowest-priority substituent of the four is pointed away from the viewer, the viewer will then see two possibilities: if the priority of the remaining three substituents decreases in clockwise direction, it is labeled R (for Latin: rectus -right); if it decreases in counterclockwise direction, it is S (for Latin: sinister -left).
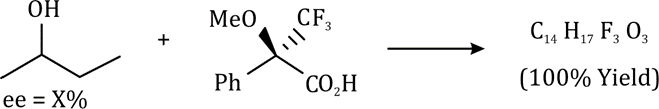
In stereochemistry, prochiral molecules are those that can be converted from achiral to chiral in a single step molecule by replacing a single atom or group. Prochiral centers are often found in organic molecules where certain atoms or groups are related by symmetry.
Determining Prochirality: To assign pro-R or pro-S, replace one of the identical atoms (like H) with a hypothetical atom/group of higher priority. An atom with higher atomic number gets higher priority.