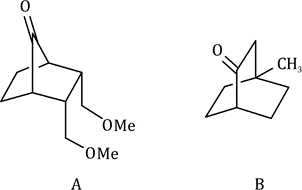
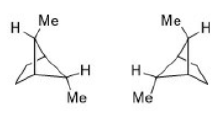
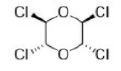
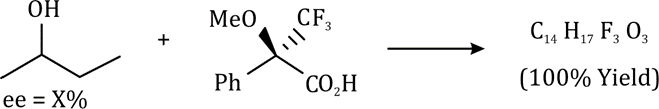
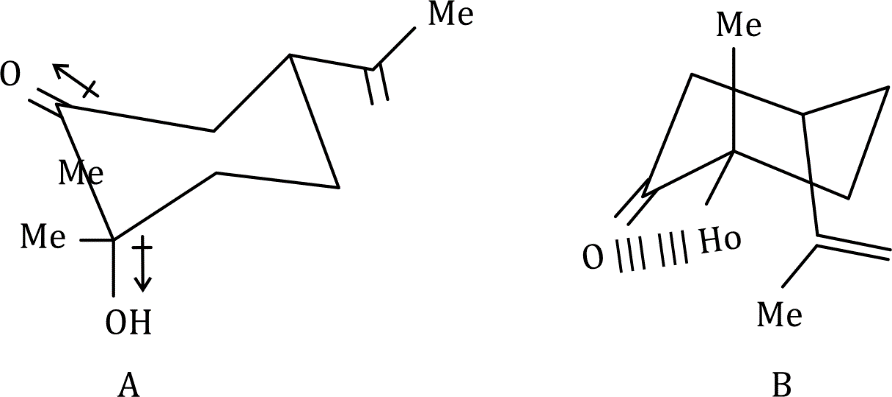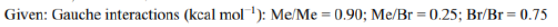Correct option is B
In chemistry, a hydrogen bond (or H-bond) is primarily an electrostatic force of attraction between a hydrogen (H) atom which is covalently bonded to a more electronegative "donor" atom or group, and another electronegative atom bearing a lone pair of electrons-the hydrogen bond acceptor. Hydrogen bonds can be intermolecular (occurring between separate molecules) or intramolecular (occurring among parts of the same molecule).
Dimethyl sulfoxide (DMSO) is an organosulfur compound with the formula (CH3)2SO. This colorless liquid is the sulfoxide most widely used commercially. It is an important polar aprotic solvent.
2,2,4-Trimethylpentane, also known as isooctane, is an organic compound with the formula (CH3)3CCH2CH(CH3)2. It is one of several isomers of octane (C8H18). Isooctane is a very good solvent for nonpolar substances.
Dipoles generally occur between two non-metals that share electrons as part of their bond. Since each atom has a different affinity for electrons, the ‘push and pull’ of their shared electrons results in one atom maintaining most of the electron density and a partial negative charge, leaving the other atom with a partial positive charge.
A dipole moment is the product of the magnitude of the charge and the distance between the centers of the positive and negative charges. In chemistry, the arrows that are drawn in order to represent dipole moments begin at the positive charge and end at the negative charge.
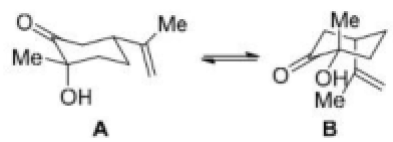
There will be intramolecular hydrogen bonding in conformation B. High number of DMSO molecules will solvate B and create stearic crowding. Hence, B will be more stable in isooctane.
A will predominate in DMSO due to opposing dipole interaction. DMSO can also participate in intermolecular hydrogen bonding in A.