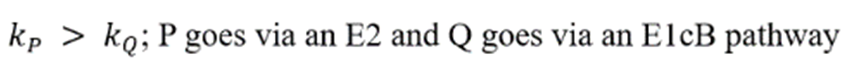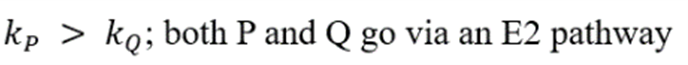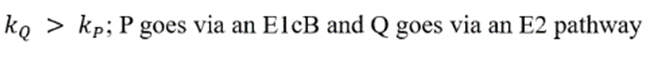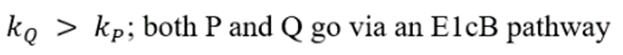Correct option is B
The E2 mechanism, where E2 stands for bimolecular elimination, involves a one-step mechanism in which carbon-hydrogen and carbon-halogen bonds break to form a double bond.
The specifics of the reaction are as follows:
1.E2 is a single step elimination, with a single transition state.
2.The reaction rate is second order, because it's influenced by both the alkyl halide and the base (bimolecular).
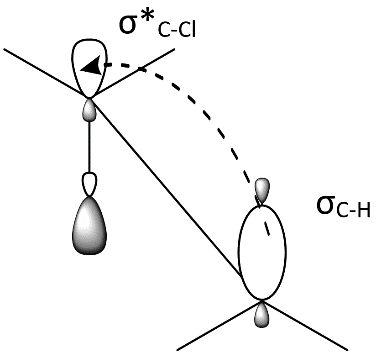
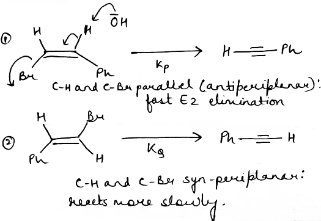
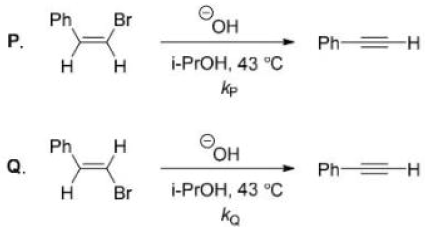
3.Because the E2 mechanism results in the formation of a pi bond, the two leaving groups (often a hydrogen and a halogen) need to be antiperiplanar. Antiperiplanar describes that the molecule will be in a staggered arrangement with the anti-periplanar functional groups pointing up and down, 180° away from each other. An antiperiplanar transition state has staggered conformation with lower energy than a synperiplanar transition state which is in eclipsed conformation with higher energy. The reaction mechanism involving staggered conformation is more favorable for E2 reactions.
4.E2 typically uses a strong base. It must be strong enough to remove a weakly acidic hydrogen.