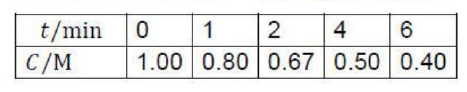
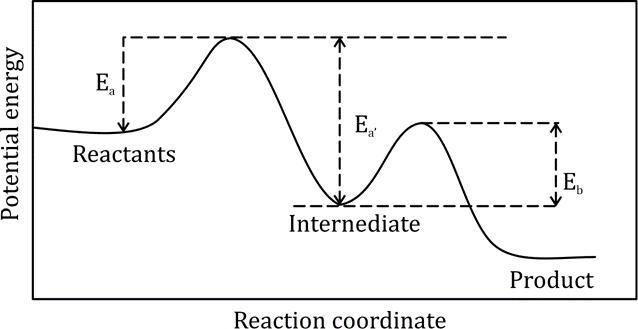
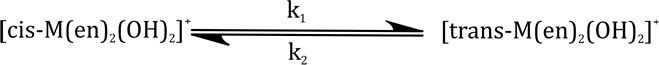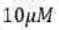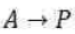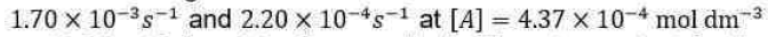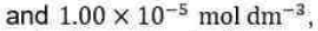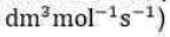Correct option is B
Arrhenius proposed the following empirical equation for calculating the energy of activation of a reaction having rate constant k at temperature T:
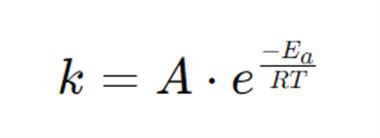
where Ea is called the Arrhenius activation energy and A is called the Arrhenius pre-exponential factor. Since the exponential factor is dimensionless, the pre-exponential factor A has the same units as the rate constant k. The units of k for a first-order reaction are s-1, which is the unit of frequency. Hence, A is also called the frequency factor. Ea, and A are called the Arrhenius parameters.