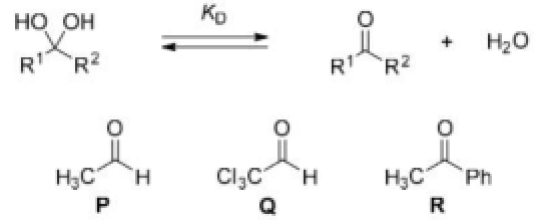
Correct option is B
The three chlorine atoms present in Q make the carbonyl carbon more electron deficient. Hence, the tendency of attack by water on the electron deficient carbonyl carbon will be highly favored.
The carbonyl carbon in acetaldehyde (P) is attached to a hydrogen atom and a methyl group (CH₃). The methyl group is slightly electron-donating due to its +I effect, but the hydrogen contributes nothing in terms of electron donation. As a result, the carbonyl carbon remains relatively electron-deficient.
The carbonyl carbon in acetophenone (R) is attached to a phenyl group (C₆H₅) and a methyl group (CH₃). The phenyl group is a conjugated system and can donate electron density to the carbonyl group via resonance. This resonance effect reduces the electron deficiency of the carbonyl carbon.
Therefore, the backward reaction follows the order: Q>P>R