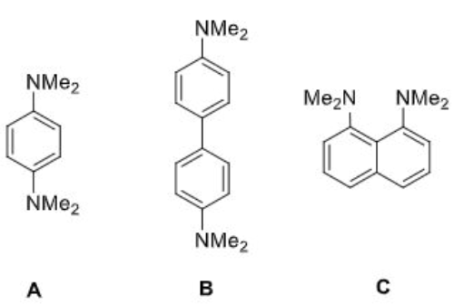
Correct option is D
Basicity of amines can be defined as their capacity to donate the lone pair of electrons on the N atom. This makes the amines’ basic’ as they can easily donate that pair to an electron-deficient centre.
1,8-Bis(dimethylamino)naphthalene is an organic compound with the formula C10H6(NMe2)2 (Me = methyl). It is classified as a peri-naphthalene, i.e. a 1,8-disubstituted derivative of naphthalene. Owing to its unusual structure, it exhibits exceptional basicity. It is often referred by the trade name Proton Sponge, a trademark of Sigma-Aldrich.
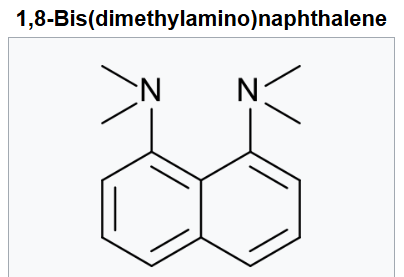
This compound is a diamine in which the two dimethylamino groups are attached on the same side (peri position) of a naphthalene ring. This molecule has several very interesting properties; one is its very high basicity; another is its spectroscopic properties.
The high basicity is attributed to the relief of strain upon protonation and/or the strong interaction between the nitrogen lone pairs. Additionally, although many aromatic amines such as aniline show reduced basicity (due to nitrogen being sp2 hybridized; its lone pair occupying a 2p orbital and interacting and being withdrawn by the aromatic ring), this is not possible in this molecule, as the nitrogens' methyl groups prevent its substituents from adopting a planar geometry, as this would require forcing methyl groups from each nitrogen atom into one another—thus the basicity is not reduced by this factor which is found in other molecules.
A is more basic than B. The electron delocalization of the nitrogen lone pair will be more in B (due to the presence of two rings) than in A. Thus, the electrons will be less available for donation to an electrophilic centre in B.
Additional Knowledge
In chemistry, the mesomeric effect (or resonance effect) is a property of substituents or functional groups in a chemical compound. It is defined as the polarity produced in the molecule by the interaction of two pi bonds or between a pi bond and lone pair of electrons present on an adjacent atom. This change in electron arrangement results in the formation of resonance structures that hybridize into the molecule's true structure. The pi electrons then move away from or toward a particular substituent group.
The +M effect, also known as the positive mesomeric effect, occurs when the substituent is an electron donating group. The group must have one of two things: a lone pair of electrons, or a negative charge. In the +M effect, the pi electrons are transferred from the group towards the conjugate system, increasing the density of the system. Due to the increase in electron density, the conjugate system will develop a more negative charge. As a result, the system under the +M effect will be more reactive towards electrophiles, which can take away the negative charge, than a nucleophile.
+M effect order: