Correct option is A
Molecules that bind to a specific substance inside or on the surface of it and cause a specific effect are known as receptors. Receptors can bind to a specific ligand through strong H-bonding interaction and can form a hydrogen-bonded complex.
The strength of an H-bonded complex depends upon neighboring donor and acceptor sites. The more the number of H bonding more will be the stability of the complex.
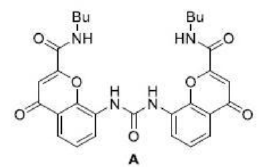
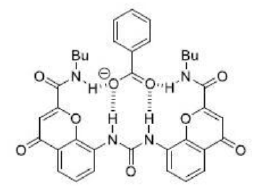
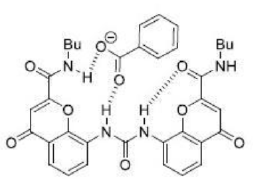
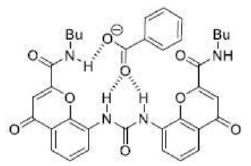
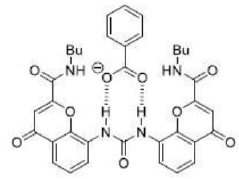
The benzoate anion acts as a ligand through two electronegative O atoms and perfectly fits into the cavity of receptor A. The four H atom present inside the periphery of receptor A results in a strong H-bonded complex with four H bonds.
A hydrogen bond (H-bond) is a specific type of molecular interaction that exhibits partial covalent character and cannot be described as a purely electrostatic force. It occurs when a hydrogen (H) atom, covalently bonded to a more electronegative donor atom or group (Dn), interacts with another electronegative atom bearing a lone pair of electrons—the hydrogen bond acceptor (Ac).
The general notation for hydrogen bonding is Dn−H···Ac, where the solid line represents a polar covalent bond, and the dotted or dashed line indicates the hydrogen bond. The most frequent donor and acceptor atoms are nitrogen (N), oxygen (O), and fluorine (F), due to their high electronegativity and ability to engage in stronger hydrogen bonding.