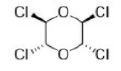
Correct option is A
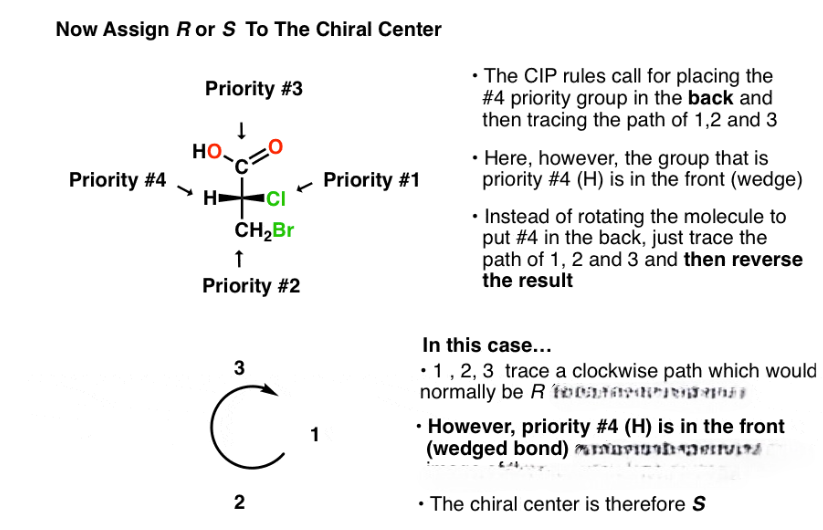
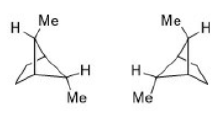
The R/ S system is an important nomenclature system for denoting enantiomers. This approach labels each chiral center R or S according to a system by which its substituents are each assigned a priority, according to the Cahn–Ingold–Prelog priority rules (CIP), based on atomic number. When the center is oriented so that the lowest-priority substituent of the four is pointed away from the viewer, the viewer will then see two possibilities: if the priority of the remaining three substituents decreases in clockwise direction, it is labeled R (for Latin: rectus – right); if it decreases in counterclockwise direction, it is S (for Latin: sinister – left).
The Cahn–Ingold–Prelog (CIP) sequence rules are a standard process to completely and unequivocally name a stereoisomer of a molecule. The purpose of the CIP system is to assign an R or S descriptor to each stereocenter.
1. Compare the atomic number ( Z) of the atoms directly attached to the stereocenter; the group having the atom of higher atomic number Z receives higher priority (i.e. number 1).
2. If there is a tie, the atoms at distance 2 from the stereocenter have to be considered: a list is made for each group of further atoms bonded to the one directly attached to the stereocenter. Each list is arranged in order of decreasing atomic number Z. Then the lists are compared atom by atom; at the earliest difference, the group containing the atom of higher atomic number Z receives higher priority.
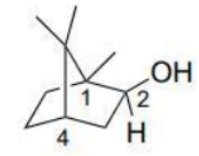
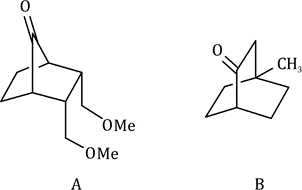
Application in Bicyclic Systems
· In bicyclic compounds, stereochemical descriptors (R or S) are assigned to chiral centers considering the three-dimensional arrangement.
· When evaluating priorities, the rigid structure of bicyclic compounds ensures that substituents are fixed in specific spatial orientations, aiding in the determination of configurations.
Carbon number 1: The highest priority is assigned to the carbon which has an OH group attached to it. Second priority goes to the carbon which is further attached to three carbons. Third priority should be given to the carbon which is attached to one carbon atom and two hydrogen atoms. Lastly, methyl group gets the fourth priority. Thus, the configuration is S.
Carbon number 2: First priority is assigned to the OH group. Second priority goes to the carbon which is further attached to three carbons. Third priority should be given to the carbon which is attached to one carbon atom and two hydrogen atoms. Lastly, hydrogen atom gets the fourth priority. Thus, the configuration is S.
Carbon number 4: The highest priority goes to the carbon which is further attached to three carbons. Second priority is assigned to the carbon which is attached to one carbon (which has an OH group attached to it) and two hydrogen atoms. Third priority goes to the carbon which is attached to one carbon atom and two hydrogen atoms. Hydrogen atom gets the fourth priority. As the hydrogen atom is coming towards the observer, the configuration changes from R to S.

Information Booster