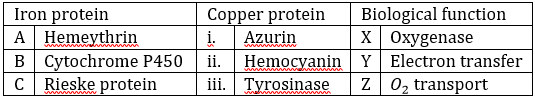
Correct option is A
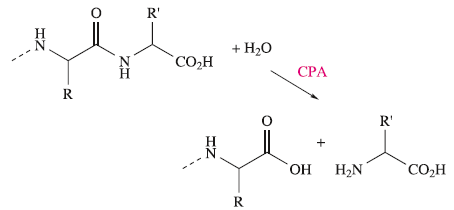
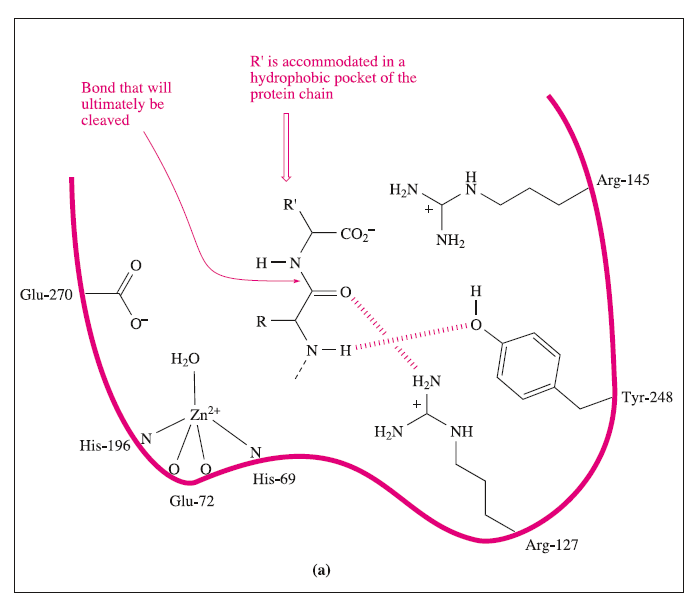
Carboxypeptidase A (CPA) is a pancreatic metalloenzyme which catalyses the cleavage of a peptide link in a polypeptide chain. The site of cleavage is specific in two ways: it occurs at the C-terminal amino acid, and it exhibits a high selectivity for substrates in which the C- terminal amino acid contains a large aliphatic or Ph substituent. The latter arises from the presence, near to the active site, of a hydrophobic pocket in the protein which is compatible with the accommodation of, for example, a Ph group
Carboxypeptidase A is monomeric
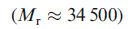
and exists in three forms
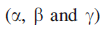
which contain 307, 305 and 300 amino acids respectively. Near the surface of the protein lies a pocket in which a Zn2+ion is bound to the protein backbone by one bidentate Glu and two His residues. A 5-coordinate coordination sphere is completed by a water molecule
In the first step, the peptide to be cleaved is ‘manoeuvred’ into position close to the Zn2+ site; the dominant substrate–protein interactions involved at this stage are:

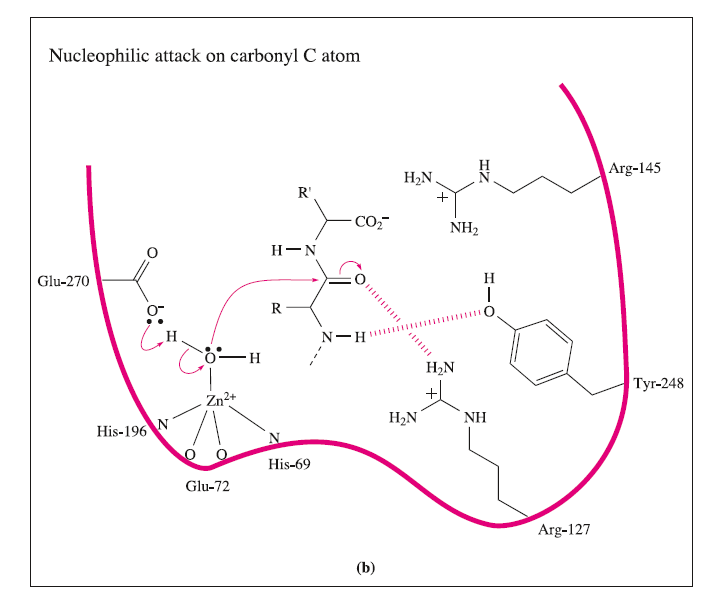
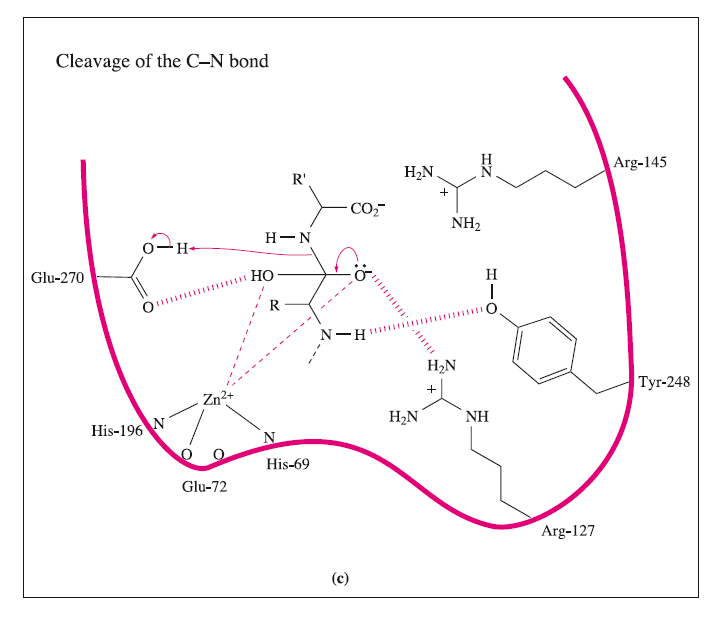
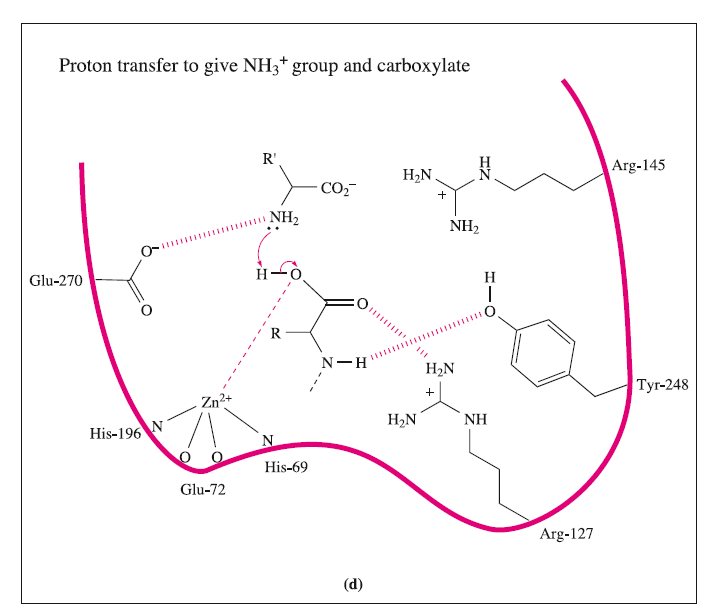
These interactions may be supplemented by hydrogen bond formation between the OH group of Tyr-248 and the N-H group indicated in the figure, and between Arg-127 and the C=O group adjacent to the peptide cleavage site. This latter interaction polarizes the carbonyl group, activating it towards nucleophilic attack. The nucleophile is the H2O ligand coordinated to Zn2+.The Lewis acidity of the metal ion polarizes the O-H bonds, and (although this is not a unique proposal) it is likely that the carboxylate group of Glu-270 assists in the process by removing H+ from the H2O ligand. The cleavage of the peptide C-N bond for which H+ is probably provided by Glu-270. It appears likely that the second H+ required for the formation of the [NH3]+ group on the departing terminal amino acid comes from the terminal CO2H group of the remaining portion of the substrate.Glu-72 bound in a monodentate manner to the Zn2+centre, whereas in the rest state, a bidentate mode has been confirmed. A change from a bi- to monodentate coordination appears to be associated with the formation of the
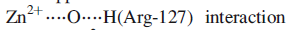
the Zn2+ ion being able to move towards Arg-127 as the interaction develops. To complete the catalytic cycle, an H2O ligand refills the vacant site on the Zn2+ centre.
A practical disadvantage of working with metalloproteins containing Zn2+ is the d10 configuration of the ion. The metal site cannot be probed by using UV-VIS or EPR spectroscopies or by magnetic measurements. Such methods were especially important before protein crystallography became a widely applied technique. Studies involving Co2+-for-Zn2+ substitution provide a metal centre that is amenable to investigation by spectroscopic and magnetic techniques (Co2+ is a d7 ion), the choice of Co2+ being because:

The Zn2+ ion can be removed from carboxypeptidase A by treatment with bipyridine, and after insertion of Co2+ , the model metalloenzyme [PCo2+] is found to be active (actually more so than native carboxypeptidase A) with respect to peptide cleavage.