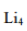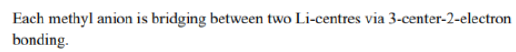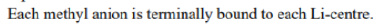Correct option is B
In the solid-state structure of methyl lithium (Li₄), each methyl anion (CH₃⁻) binds to three lithium centers via 4-center-2-electron bonding. This type of bonding involves the sharing of two electrons between four atoms, which includes two lithium atoms and the carbon atom from the methyl group. This bonding arrangement leads to a stable tetrameric structure in which each methyl group bridges between three lithium atoms.
Information Booster
Methyllithium is the simplest organolithium reagent, with the empirical formula LiCH3. This s-block organometallic compound adopts an oligomeric structure both in solution and in the solid state.
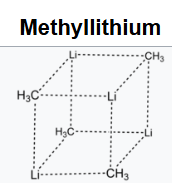
The tetrameric structure is a distorted cubane-type cluster, with carbon and lithium atoms at alternate corners. The hexameric form features hexagonal prisms with Li and C atoms again at alternate corners.
These clusters are considered "electron-deficient," that is, they do not follow the octet rule because the molecules lack sufficient electrons to form four 2-centered, 2-electron bonds around each carbon atom, in contrast to most organic compounds. The hexamer is a 30 electron compound (30 valence electrons.) If one allocates 18 electrons for the strong C-H bonds, 12 electrons remain for Li-C and Li-Li bonding. There are six electrons for six metal-metal bonds and one electron per methyl-η3 lithium interaction.