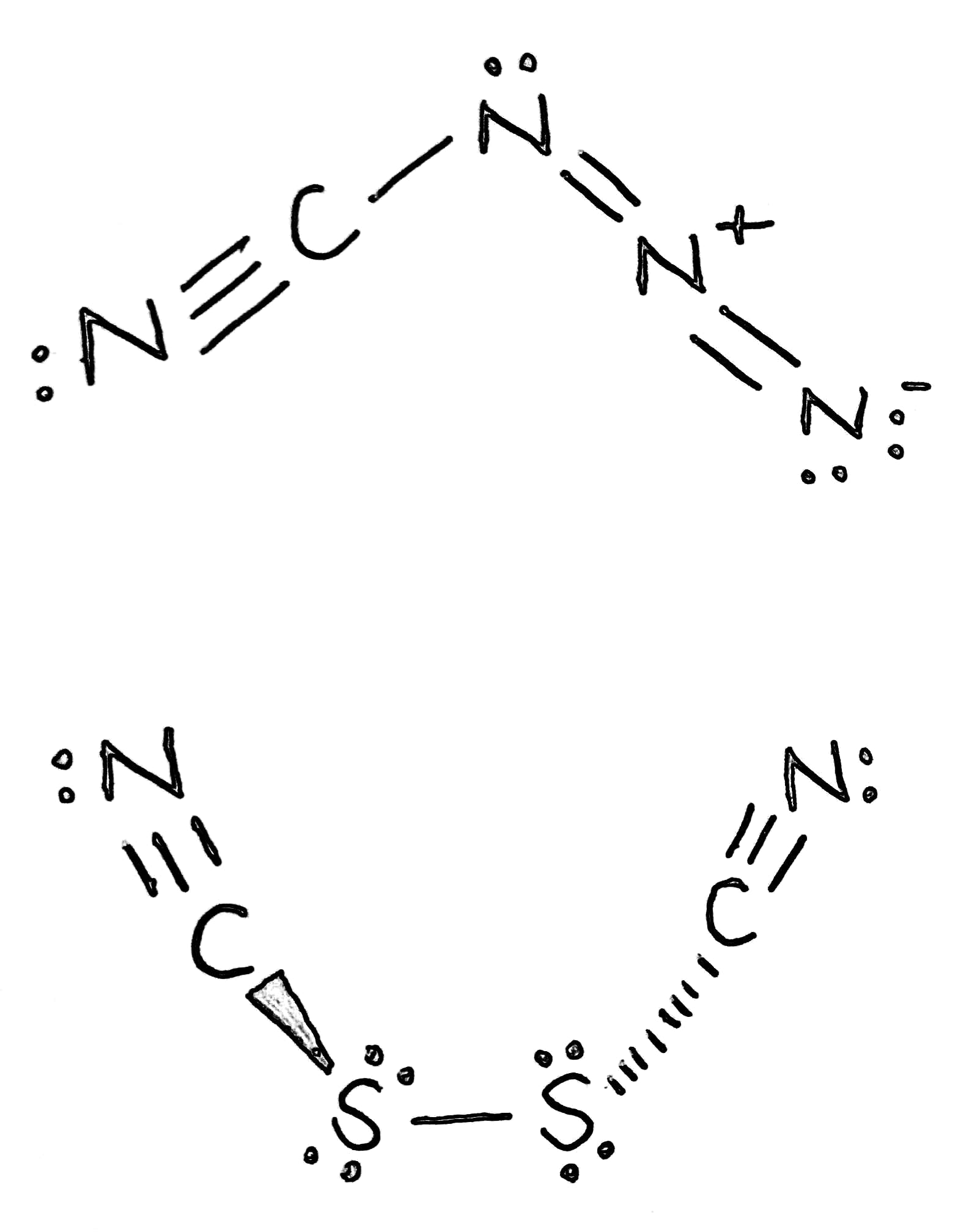
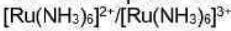Correct option is A
Cyanogen azide is a chemical compound with the chemical formula CN4, or more precisely
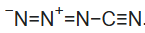
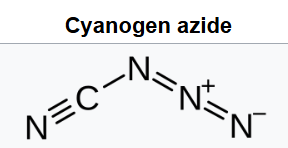
It is an azide compound of carbon and nitrogen. It is an oily, colorless liquid at room temperature. It is a highly explosive chemical that is soluble in most organic solvents, and normally handled in dilute solution in this form.
Thiocyanogen, (SCN)2, is a pseudohalogen derived from the pseudohalide thiocyanate, [SCN]-, with behavior intermediate between dibromine and diiodine.
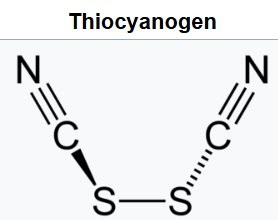
Nitrogen has five electrons in its valence shell.
Sulfur has six electrons in its valence shell.
Carbon has four electrons in its valence shell.