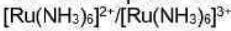Correct option is A
In this type of reaction, both complexes participating in the reaction undergo substitution reactions more slowly than the rate of electron transfer. The oxidant and the reductant come as close to each other as possible, and the coordination spheres stay intact. The transfer of an electron takes place from the reductant to the oxidant. Thus, an outer sphere mechanism involves electron transfer from the reductant to the oxidant when the intact coordination spheres are in contact at their outer edges, i.e., the distance between two metals is minimal.
An outer sphere electron transfer may occur in the following elementary steps:
In the first step, the oxidant and reductant come closer and form a precursor complex:

In the second step, there is activation of the precursor complex, which includes reorganization of the solvent molecules and changes in M–L bond lengths, occurring before electron transfer. Then, the electron transfer takes place:

In the final step, the ion pair is dissociated into products:

Salient Features of the Outer Sphere Mechanism
1.Both the oxidant and reductant should be kinetically inert.
2.The electron transfer is very fast when both the complexes are low spin including that the electron transfer takes place from t2g (π*) of reductant to the t2g (π*) of oxidant. The first reason is that energy levels of these two t2g orbitals are same. The t2g orbitals are not shielded from the ligands and the electron transfer from and to is easier and no input energy is required. The second reason is that there is no appreciable change in M-L bond length due to π*- π* electron transfer.

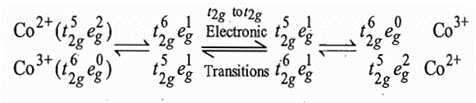
[Co (NH3)6]3+ is low spin (t2g6eg0) and [Co(NH3)6]2+ is high spin (t2g5eg2). Low spin [Co(NH3)6]3+ has an electronic configuration t2g6 with all the metal d-electrons pointing in between the ligands. On the other hand, the high spin [Co(NH3)6]2+ has an electronic configuration t2g5eg2 with two eg electrons pointing directly at the ligands. The electrons present in eg orbitals cause more repulsion with the ligands than that of t2g electrons. Therefore, Co2+-NH3 bond distance is larger than that of Co3+-NH3. The Co3+-NH3, bond distance is 193.6 pm and the Co2+-NH3 bond distance is 211.4 pm. In this case M-L bond lengths are different enough and more activation energy is needed to make them the same in the transition state.
Since Co(II) and Co(III) complexes are high spin and low spin respectively, no simple addition or removal of an electron can convert these configurations into one another. Therefore, it is necessary to excite the oxidation states before the reaction can occur as shown below: