Correct option is B
Molecular orbitals are classified as σ, π, or δaccording to their rotational symmetry about the internuclear axis, and (in centrosymmetric species) as g or u according to their symmetry with respect to inversion.
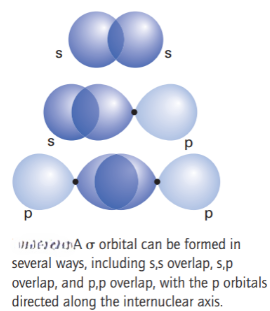
σ orbitals are formed by allowing overlap between atomic orbitals that have cylindrical symmetry around the internuclear axis, which is conventionally labelled z. The notation σ signifies that the orbital has cylindrical symmetry; atomic orbitals that can form σ orbitals include the 2s and 2pzorbitals on the two atoms. From these four orbitals (the 2s and the 2pzorbitals on atom A and the corresponding orbitals on atom B) with cylindrical symmetry we can construct four σ molecular orbitals, two of which arise predominantly from interaction of the 2s orbitals, and two from interaction of the 2pzorbitals. These molecular orbitals are labelled 1σg , 1σu , 2σg , and 2σu , respectively. Interaction between a 2s on one atom and a 2pzorbital on the other atom is possible if their relative energies are similar.
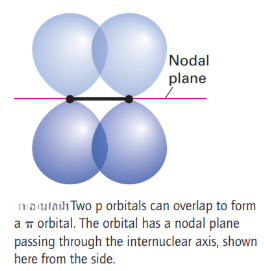
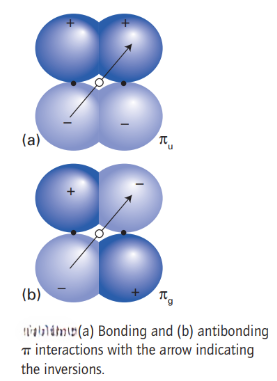
The remaining two 2p orbitals on each atom, which have a nodal plane containing the z-axis, overlap to give π orbitals. Bonding and antibonding π orbitals can be formed from the mutual overlap of the two 2pxorbitals, and also from the mutual overlap of the two 2pyorbitals. This pattern of overlap gives rise to the two pairs of doubly degenerate energy levels (two energy levels of the same energy) and labelled 1πu and 1πg.
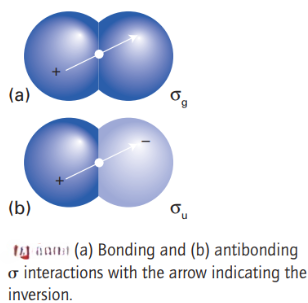
For homonuclear diatomics, it is sometimes convenient to signify the symmetry of the molecular orbitals with respect to their behaviour under inversion through the centre of the molecule. The operation of inversion consists of starting at an arbitrary point in the molecule, travelling in a straight line to the centre of the molecule, and then continuing an equal distance out on the other side of the centre. The orbital is designated g (for gerade, even) if it is identical under inversion, and u (for ungerade, odd) if it changes sign. Thus, a bonding σ orbital is g and an antibonding σ orbital is u. On the other hand, a bonding π orbital is u and an antibonding π orbital is g.
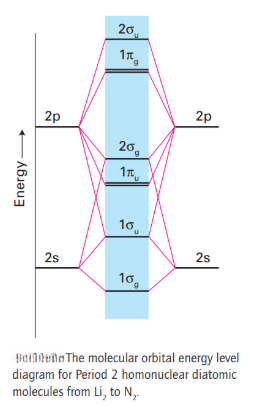
The second period contains the elements lithium, beryllium, boron, carbon, nitrogen, oxygen, fluorine, and neon.
For O2 and F2 the order of the σ and π orbitals is reversed.
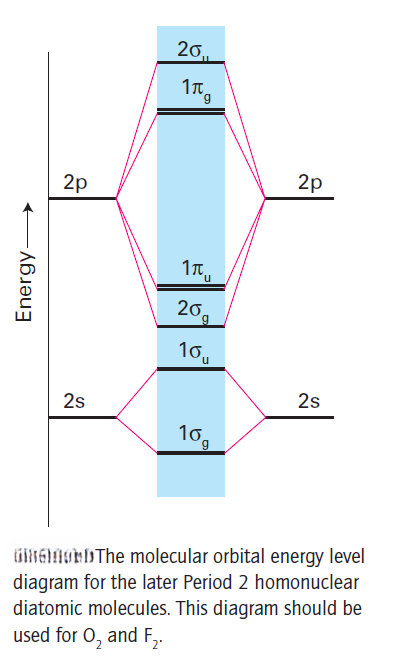
The reversal of order can be traced to the increasing separation of the 2s and 2p orbitals that occurs on going to the right across Period 2. A general principle of quantum mechanics is that the mixing of wavefunctions is strongest if their energies are similar; mixing is not important if their energies differ by more than about 1 eV. When the s, p energy separation is small, each σ molecular orbital is a mixture of s and p character on each atom. As the s and p energy separation increases, the molecular orbitals become more purely s-like and p-like.