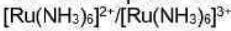Correct option is B
In physics and chemistry, ionization energy (IE) is the minimum energy required to remove the most loosely bound electron of an isolated gaseous atom, positive ion, or molecule. The first ionization energy is quantitatively expressed as
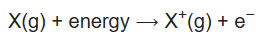
where X is any atom or molecule, X+ is the resultant ion when the original atom was stripped of a single electron, and e- is the removed electron. Roughly speaking, the closer the outermost electrons are to the nucleus of the atom, the higher the atom's ionization energy.
Comparison of ionization energies of atoms in the periodic table reveals two periodic trends which follow the rules of Coulombic attraction:
Ionization energy generally increases from left to right within a given period (that is, row).
Ionization energy generally decreases from top to bottom in a given group (that is, column).
The latter trend results from the outer electron shell being progressively farther from the nucleus, with the addition of one inner shell per row as one moves down the column.
The nth ionization energy refers to the amount of energy required to remove the most loosely bound electron from the species having a positive charge of (n − 1). For example, the first three ionization energies are defined as follows:
1st ionization energy is the energy that enables the reaction
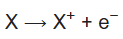
2nd ionization energy is the energy that enables the reaction
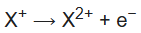
3rd ionization energy is the energy that enables the reaction
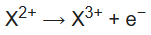
Ionization energy is also a periodic trend within the periodic table. Moving left to right within a period, or upward within a group, the first ionization energy generally increases, with exceptions such as aluminium and sulfur in the table above. As the nuclear charge of the nucleus increases across the period, the electrostatic attraction increases between electrons and protons, hence the atomic radius decreases, and the electron cloud comes closer to the nucleus because the electrons, especially the outermost one, are held more tightly by the higher effective nuclear charge.
On moving downward within a given group, the electrons are held in higher-energy shells with higher principal quantum number n, further from the nucleus and therefore are more loosely bound so that the ionization energy decreases. The effective nuclear charge increases only slowly so that its effect is outweighed by the increase in n.
The boron group are the chemical elements in group 13 of the periodic table, consisting of boron (B), aluminium (Al), gallium (Ga), indium (In) and thallium (Tl).
Scandium belongs to group 3 while cobalt belongs to group 9.
Selenium belongs to group 16.
There are exceptions to the general trend of rising ionization energies within a period. For example, the value decreases from (7N : 14.5 eV) to oxygen (8O : 13.6 eV). This dip can be explained in terms of electron configurations.
In oxygen, the last electron shares a doubly occupied p-orbital with an electron of opposing spin. The two electrons in the same orbital are closer together on average than two electrons in different orbitals, so that they shield each other from the nucleus more effectively and it is easier to remove one electron, resulting in a lower ionization energy.