Correct option is C
Valence shell electron pair repulsion (VSEPR) theory is a model used in chemistry to predict the geometry of individual molecules from the number of electron pairs surrounding their central atoms. The premise of VSEPR is that the valence electron pairs surrounding an atom tend to repel each other. The greater the repulsion, the higher in energy (less stable) the molecule is. Therefore, the VSEPR-predicted molecular geometry of a molecule is the one that has as little of this repulsion as possible.
VSEPR theory is used to predict the arrangement of electron pairs around central atoms in molecules, especially simple and symmetric molecules. A central atom is defined in this theory as an atom which is bonded to two or more other atoms, while a terminal atom is bonded to only one other atom.
The overall geometry is further refined by distinguishing between bonding and nonbonding electron pairs. The bonding electron pair shared in a sigma bond with an adjacent atom lies further from the central atom than a nonbonding (lone) pair of that atom, which is held close to its positively charged nucleus. VSEPR theory therefore views repulsion by the lone pair to be greater than the repulsion by a bonding pair. As such, when a molecule has 2 interactions with different degrees of repulsion, VSEPR theory predicts the structure where lone pairs occupy positions that allow them to experience less repulsion. Lone pair–lone pair (lp–lp) repulsions are considered stronger than lone pair–bonding pair (lp–bp) repulsions, which in turn are considered stronger than bonding pair–bonding pair (bp–bp) repulsions, distinctions that then guide decisions about overall geometry when 2 or more non-equivalent positions are possible
The electronic geometry of a molecule depends on the number of bond pairs and lone pairs, while the shape depends only on the number of bond pairs.
The steric number of a central atom in a molecule is the number of atoms bonded to that central atom, called its coordination number, plus the number of lone pairs of valence electrons on the central atom.
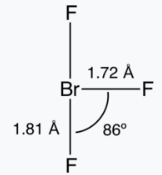
BrF3
Bromine trifluoride is an interhalogen compound with the formula BrF3. At room temperature, it is a straw-coloured liquid with a pungent odor which decomposes violently on contact with water and organic compounds.
The BrF3 molecule is T-shaped and planar. In the VSEPR formalism, the bromine center is assigned two electron lone pairs. The distance from the bromine atom to each axial fluorine atom is 1.81Å and to the equatorial fluorine atom is 1.72Å. The angle between an axial fluorine atom and the equatorial fluorine atom is slightly smaller than 90° — the 86.2° angle observed is due to the repulsion generated by the electron pairs being greater than that of the Br-F bonds.
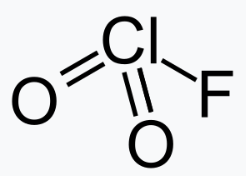
FClO2
Chloryl fluoride is the chemical compound with the formula ClO2F. It is commonly encountered as side-product in reactions of chlorine fluorides with oxygen sources. It is the acyl fluoride of chloric acid.
ClO2F is a pyramidal molecule as predicted by VSEPR. The structure reflects the greater tendency of chlorine to exist in positive oxidation states with oxygen and fluorine ligands.
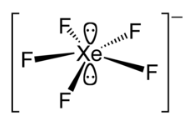
[XeF5]-
The [XeF5]- ion was the first example of a pentagonal planar molecular geometry. The anion is planar, with the fluorine atoms in a slightly distorted pentagonal coordination (Xe–F bond lengths 197.9–203.4 pm, and F–X–F bond angles 71.5°–72.3°).
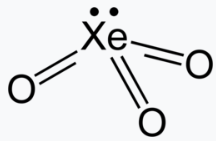
XeO3
Xenon trioxide is an unstable compound of xenon in its +6 oxidation state. It is a very powerful oxidizing agent, and liberates oxygen from water slowly, accelerated by exposure to sunlight. XeO3 has a trigonal pyramidal shape.