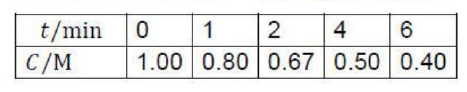
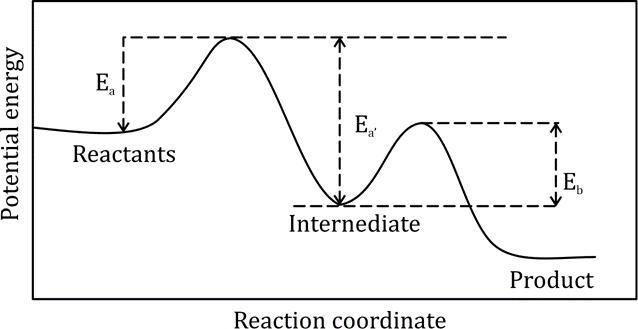
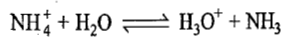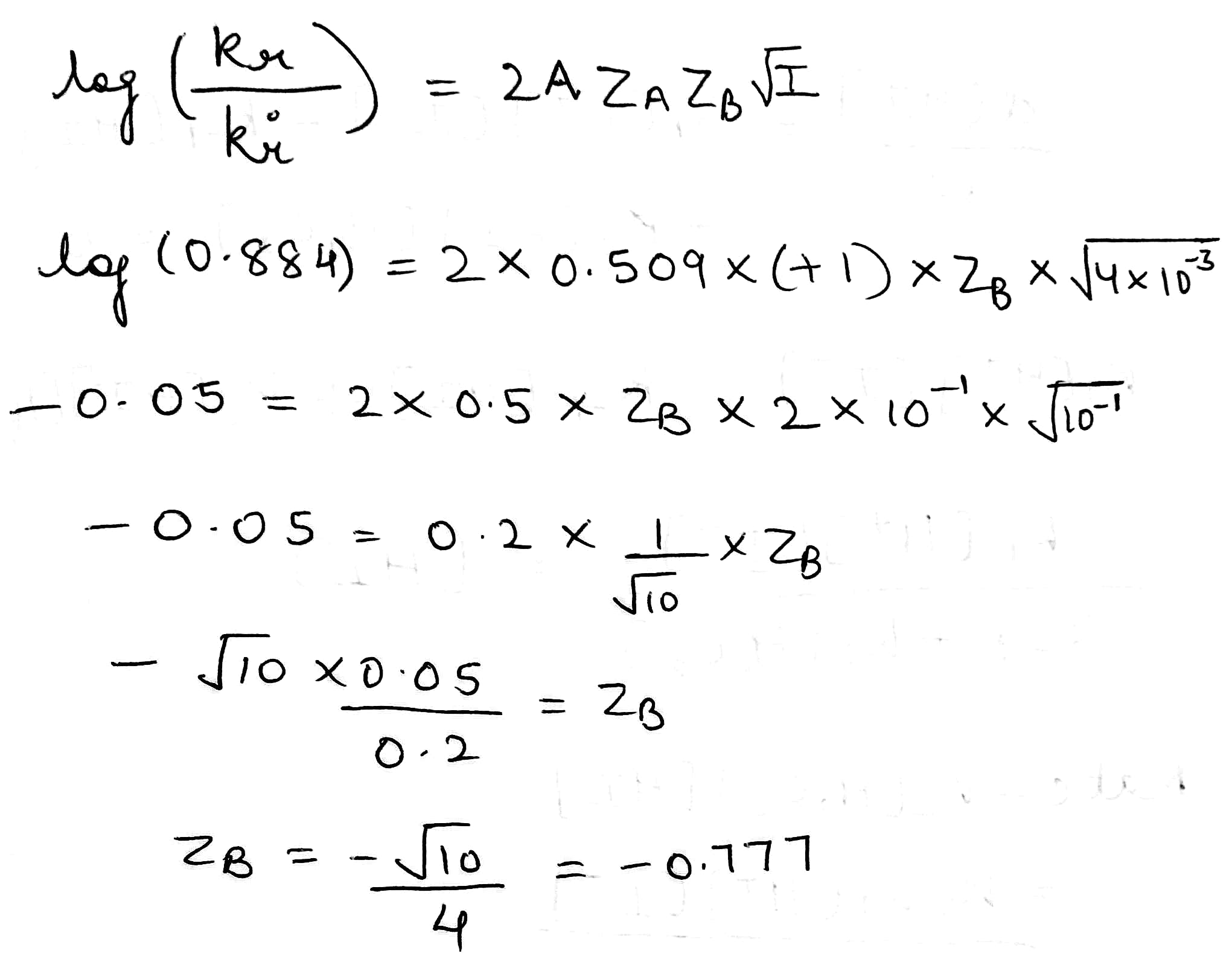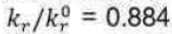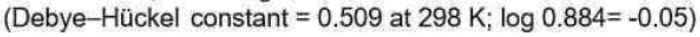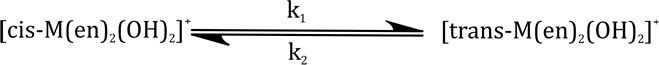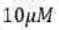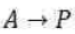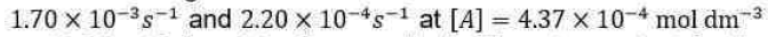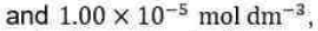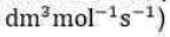Correct option is C
Effect of Ionic Strength (Primary and Secondary Salt Effects)
For an ionic reaction
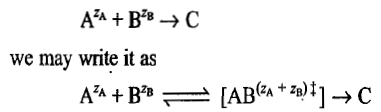
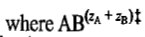
is the transition-state complex. For the equilibrium between the reactants and the activated complex, we may write

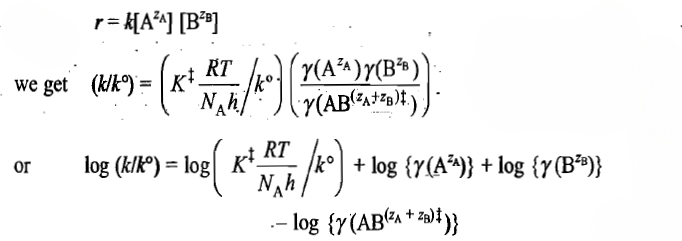
The activity coefficient of the ion depends upon the ionic strength μ of the solution. The latter is defined as


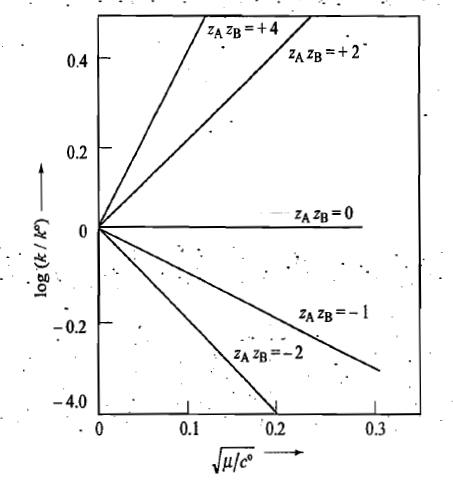
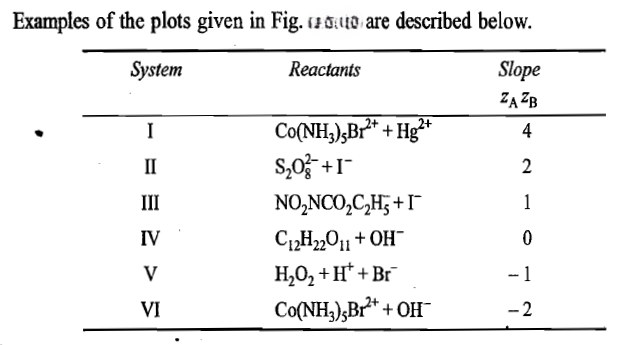
Primary Salt Effect
The change in k with ionic strength is known as the primary salt effect.

The primary salt effect is due to the change in the value of activity coefficients of reactants and intermediate complex due to the ionic strength of the solution.
Secondary Salt Effect
There is another type of salt action, known as the secondary salt effect which influences the rate of reaction by altering the effective concentration of the catalyzing species. Take, for example, the hydrolysis of cane sugar catalyzed by a weak acid. The experimental rate constant is given by

If the catalyzing acid is NH4+,H3O+ by the reaction