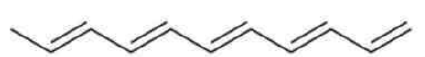
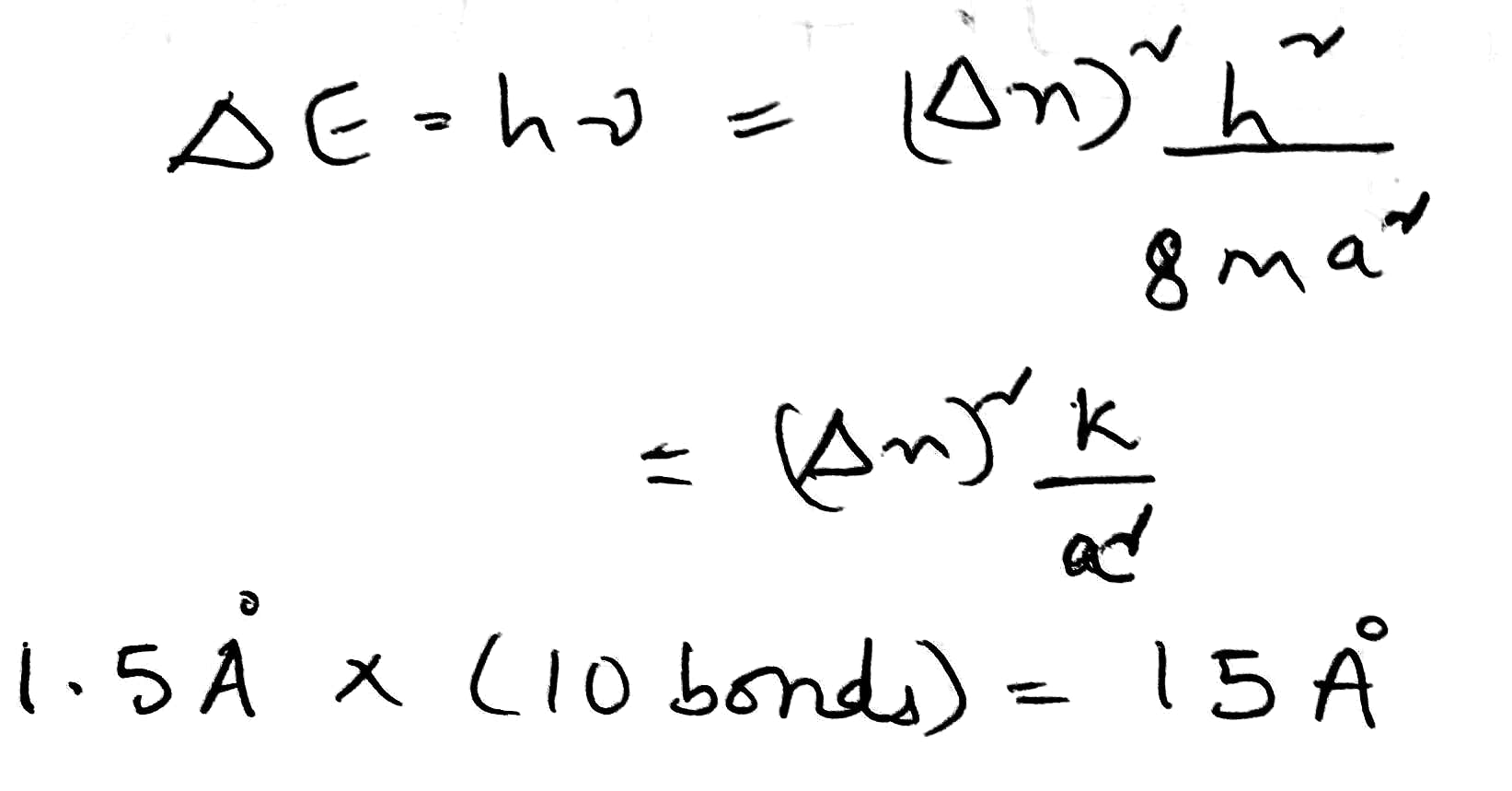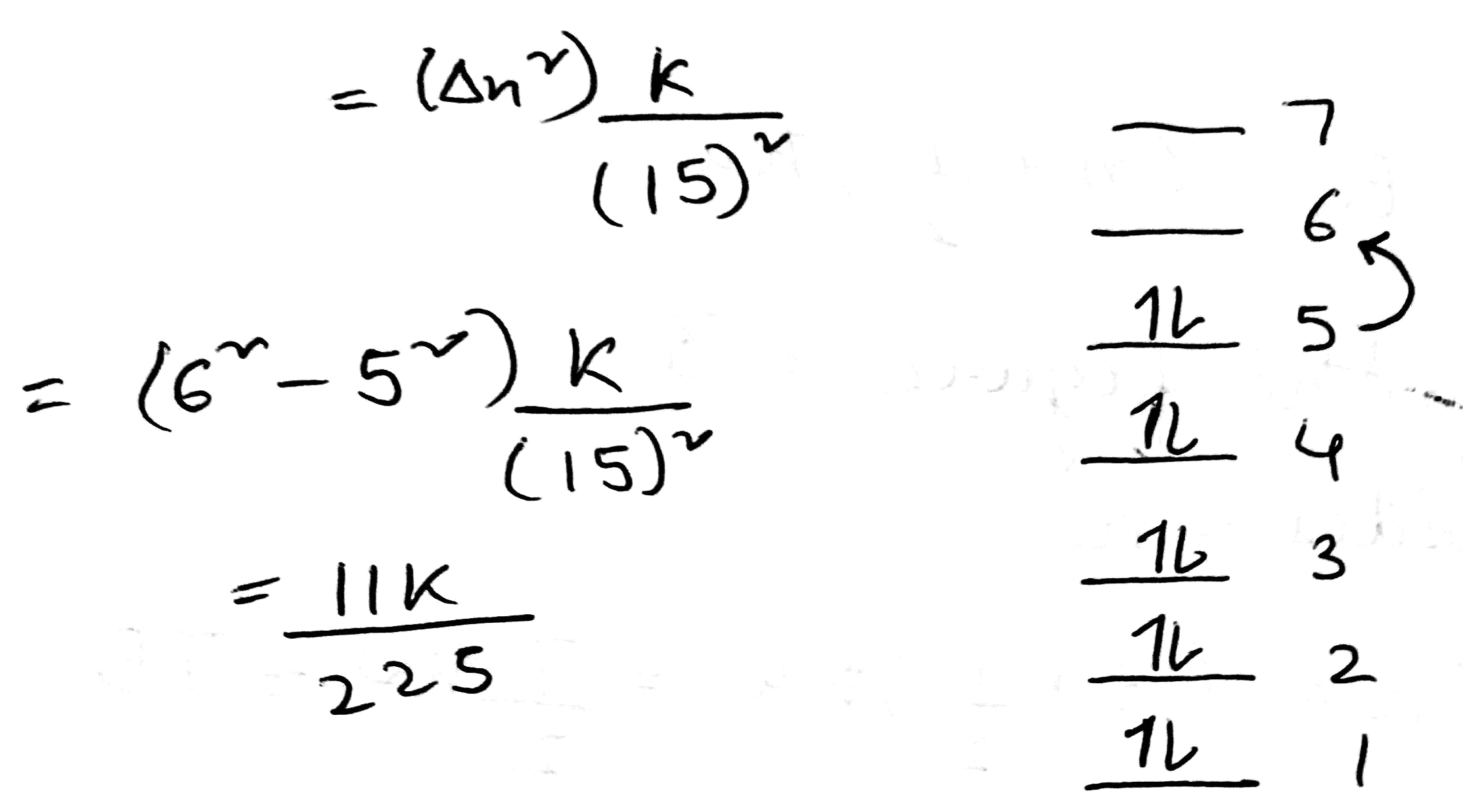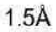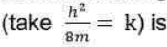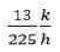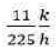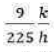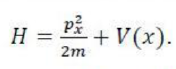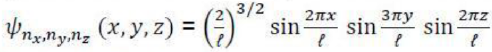Correct option is B
A particle in a box
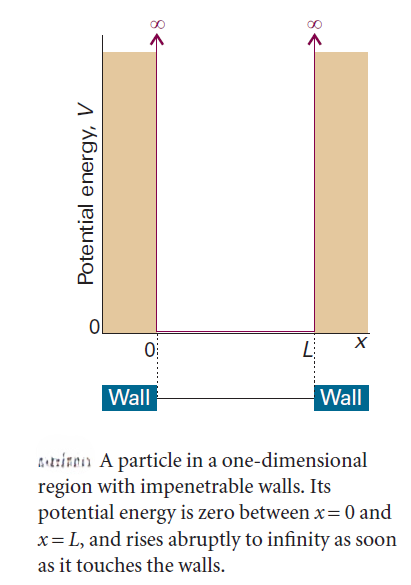
We consider a particle in a box, in which a particle of mass m is confined between two walls at x = 0 and x = L: the potential energy is zero inside the box but rises abruptly to infinity at the walls. This model is an idealization of the potential energy of a gas-phase molecule that is free to move in a one-dimensional container. However, it is also the basis of the treatment of the electronic structure of metals and of a primitive treatment of conjugated molecules. The particle in a box is also used in statistical thermodynamics in assessing the contribution of the translational motion of molecules to their thermodynamic properties.
The acceptable solutions
The Schrödinger equation is
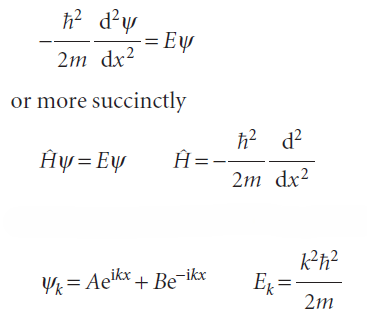
Note that we are now labelling both the wavefunctions and the energies (that is, the eigenfunctions and eigenvalues of
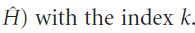
We can verify that these functions are solutions by substituting
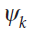
into the left-hand side of equation and showing that the result is equal to
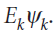
In this case, all values of k, and therefore all values of the energy, are permitted. It follows that the translational energy of a free particle is not quantized.

The Schrödinger equation for the region between the walls (where V = 0) is the same as for a free particle , so the general solutions are also the same. However, we can use
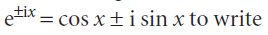
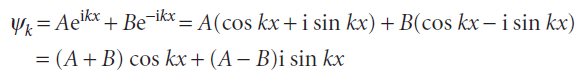
If we absorb all numerical factors into two new coefficients C and D, then the general solutions take the form
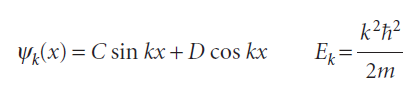
For a free particle, any value of Ek corresponds to an acceptable solution. However, when the particle is confined within a region, the acceptable wavefunctions must satisfy certain boundary conditions, or constraints on the function at certain locations. As we shall see when we discuss penetration into barriers, a wavefunction decays exponentially with distance inside a barrier, such as a wall, and the decay is infinitely fast when the potential energy is infinite. This behaviour is consistent with the fact that it is physically impossible for the particle to be found with an infinite potential energy. We conclude that the wavefunction must be zero where V is infinite,