Correct option is A
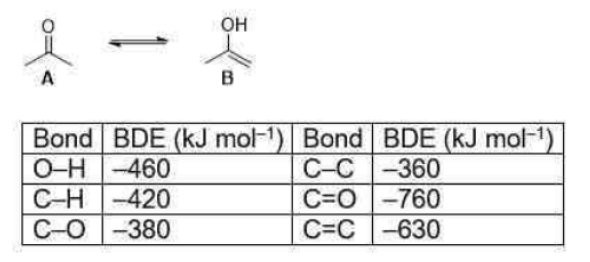
The bond-dissociation energy is one measure of the strength of a chemical bond A−B. It can be defined as the standard enthalpy change when A−B is cleaved by homolysis to give fragments A and B, which are usually radical species. The enthalpy change is temperature-dependent, and the bond-dissociation energy is often defined to be the enthalpy change of the homolysis at 0 K (absolute zero), although the enthalpy change at 298 K (standard conditions) is also a frequently encountered parameter.
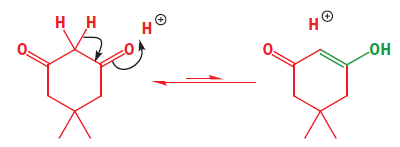
An enol is exactly what the name implies: an ene-ol. It has a C=C double bond and an OH group joined directly to it. Simple carbonyl compounds have enols too—the enol of cyclohexanone is given as:
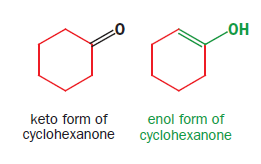
In the case of dimedone, the enol must be formed by a transfer of a proton from the central CH2 group of the keto form to one of the OH groups. Notice that there is no change in pH—a proton is lost from carbon and gained on oxygen. The reaction is known as enolization as it is the conversion of a carbonyl compound into its enol. It is a strange reaction in which little happens. The product is almost the same as the starting material since the only change is the transfer of one proton and the shift of the double bond. Reactions like this are given the name tautomerism.
Although any carbonyl compound with protons adjacent to the carbonyl group can enolize, simpler carbonyl compounds like cyclohexanone or acetone have only a trace of enol present under ordinary conditions. The equilibrium lies well over towards the keto form (the equilibrium constant K for acetone is about 10-6).
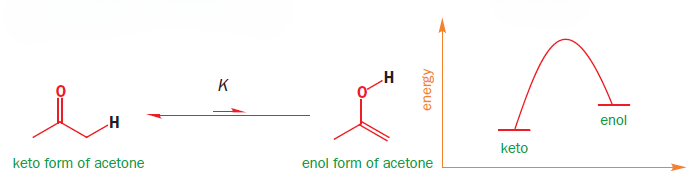
This is because the combination of a C=C double bond and an O–H single bond is (slightly) less stable than the combination of a C=O double bond and a C–H single bond. The balance between the bond energies is quite fine. On the one hand, the O–H bond in the enol is a stronger bond than the C–H bond in the ketone but, on the other hand, the C=O bond of the ketone is much more stable than the C=C bond of the enol.
In the given question,
For the keto form: C-C: 360(2)=720; C=O: 760; C-H: 420(6)=2520
Total=4000
For the enol form: O-H:460;C=C:630;C-C:360;C-H:420(5)=2100;C-O=380
Total=3930
Difference=4000-3930=70