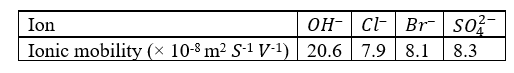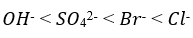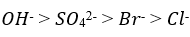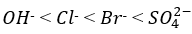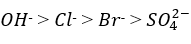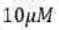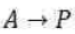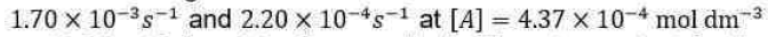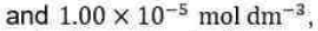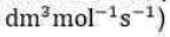Correct option is A
It is given that, chloride and bromide ions have comparatively lower ionic mobilities. This is due to the hydration of ions as a result of higher charge density around these ions because of their smaller radii. The higher charge density causes these ions to be more highly hydrated by ion-dipole interactions than the larger ions (OH- and SO42-). Since a hydrated ion has to drag along a shell of water as it moves through the solution, its mobility is naturally less than that of an unhydrated ion.