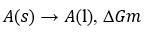Correct option is B
Consider any two phases (say, solid and liquid) of one and the same substance in equilibrium with each other at a given temperature and pressure. It is possible to transfer any definite amount of the substance from one phase to the other in a thermodynamically reversible manner, i.e., infinitesimally slowly, the system remaining in a state of equilibrium all along. For example, by supplying heat infinitesimally slowly to the system, it is possible to change any desired amount of the substance from the solid to the liquid phase at the same temperature and pressure. Similarly, by withdrawing heat infinitesimally slowly from the system, it is possible to change any desired amount of the substance from the liquid to the solid phase without any change in temperature and pressure. Since the system remains in a state of equilibrium, the free energy change of either process will be zero. Therefore, that equal amounts of a given substance must have exactly the same free energy in the two phases at equilibrium with each other.
Consider, in general, the change of a pure substance from phase A to another phase B in equilibrium with it at a given temperature and pressure. If GA is the free energy per mole of the substance in the initial phase A and GB is the free energy per mole in the final phase B, then, since GA= GB, hence, there will be no free energy change, i.e., ΔG = GB – GA = 0.