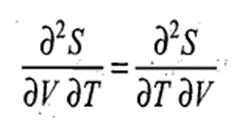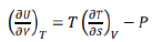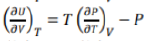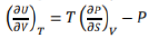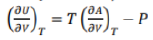Correct option is B
We can write entropy as a function of temperature and volume of the system, such that

The differential dU can be replaced in terms of dT and dV. This can be done by taking U as a function of temperature and volume.

The above equation gives the variation in entropy with volume of the system when its temperature is held constant, that is isothermal volume dependence of entropy.
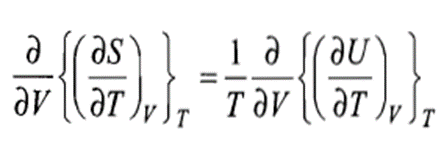
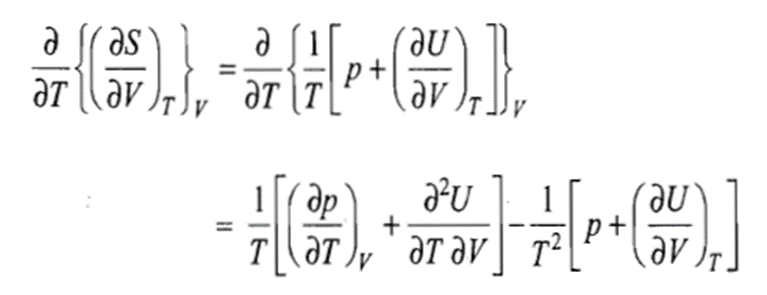
Since S is a state function, we must have