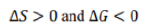Correct option is B
The changes of a system from one state to another may occur either in a reversible or in an irreversible way.
The reversible processes are characterized by the fact that when the system is restored to its original state by traversing the forward sequence of steps in the reverse order, then not only the system but also the surroundings are restored to their original states.
Operation of a Reversible Process
In a reversible process, at any stage, the external condition responsible for the process to occur differs from the internal condition by an infinitesimal amount. For example, during expansion or compression Pext=Pint ± dp and the corresponding change in volume of the system is also infinitesimally small. During this stage, the internal equilibrium is disturbed only infinitesimally and in the limit it is not disturbed at all. Thus, virtually, the system always remains in the equilibrium state during the process. Another example of reversible process is the heat exchanges between the system and the surroundings when the temperature of the latter differs from that of the former by an infinitesimal amount, i.e. Tsurr = Tsys ±dT. Obviously, a reversible process cannot be conducted in actual practice because the external condition has to differ from the internal condition by an infinitesimal amount and moreover, even if it is possible to monitor this, an infinite time would be required to complete the process. Thus, reversible processes are not real but only ideal which can be carried out only theoretically. Nevertheless, they are very important as they give the limit of the effect, whether maximum or minimum, that can be produced. Although reversible processes cannot be carried out, the goal of reversibility can be approached by adjusting the conditions with patience and skill to those of reversible processes.
At the melting point of ice (0°C, 1 atm), the system is at equilibrium. For equilibrium processes, Gibbs free energy change (ΔG) is zero because there is no net driving force. Melting involves a transition from a more ordered solid phase (ice) to a less ordered liquid phase (water). Hence, the entropy change (ΔS) is positive as the molecular randomness increases.