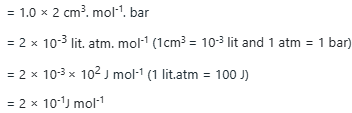Correct option is C
The molar Gibbs free energy is a thermodynamic property of a substance that describes its potential to do non-mechanical work (such as chemical reactions) at constant temperature and pressure. It is denoted by the symbol G and is usually measured in joules per mole (J/mol) or sometimes in kilojoules per mole (kJ/mol).
Mathematically, the molar Gibbs free energy is defined as:
G=H-TS
dG= dH- TdS-SdT
dG=Vdp -SdT
where
G is the molar Gibbs free energy, H is the molar enthalpy (total heat content) of the system, T is the absolute temperature (in K) and S is the molar entropy.
During phase change, the temperature of the system remains constant, dT=0
dG=Vdp
According to question, during the phase transition, at constant temperature, of a solid from one form to another, the change in molar volume, ΔVm=1cm3mol-1 is independent of pressure.
When the pressure is increased from 1bar to 3 bar,
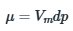
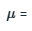
1cm3mol-1(3bar-1bar)