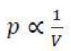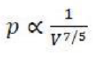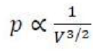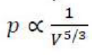Correct option is D
In adiabatic expansion or compression process heat is neither allowed to enter nor leave the system. Therefore,

If there is an expansion, w will be negative and, therefore, ΔU will also be negative, i.e. there will occur a decrease in the internal energy of the system and hence a decrease in temperature will be observed. This amounts to the fact that the work is done by the system at the expense of internal energy.If there is a compression, w will be positive and, therefore, ΔU will also be positive, i.e., there will occur an increase in the internal energy, and hence an increase in temperature will be observed. Here, the work is done by the surroundings on the system, which is stored as the internal energy.
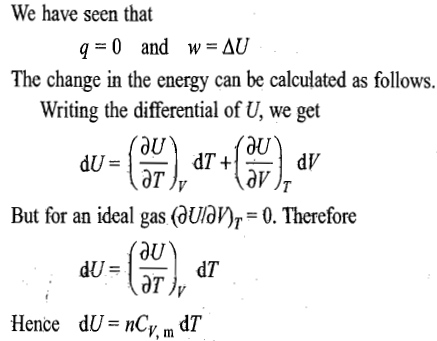
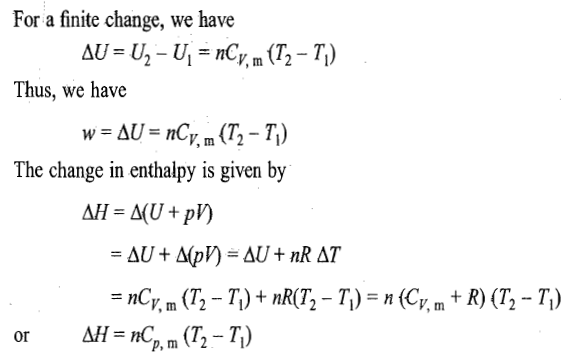
Reversible expansion or compression process
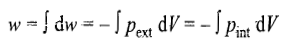
The integration in the above expression cannot be performed directly, because Pint is a function of both T and V and both variables are changing in an adiabatic expansion or compression process. However, if the final temperature is available, the values of q, w, ΔU and ΔH can be determined using the above equations. However, if the final temperature is not available, but the final pressure or volume is available, the first step is to calculate the final temperature from the expression which relates the initial and final temperatures to the respective volumes or pressures. Such expressions can be derived as follows.
Relationship between T and V
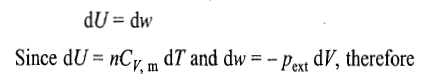


Relationship between T and P

Relationship between P and V

The heat capacity ratio is the ratio of the heat capacity at constant pressure (CP) to heat capacity at constant volume (CV). It is sometimes also known as the isentropic expansion factor and is denoted by γ (gamma) for an ideal gas.
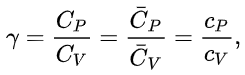
where C is the heat capacity,
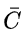
the molar heat capacity (heat capacity per mole), and c the specific heat capacity (heat capacity per unit mass) of a gas. The suffixes P and V refer to constant-pressure and constant-volume conditions respectively.
In an adiabatic process, monatomic gases have an idealised γ-factor (CP/CV) of 5/3.

Additional Knowledge
In an isothermal expansion or compression process, the temperature of the system remains constant throughout the expansion or compression process. Since for an ideal gas, U depends only on temperature
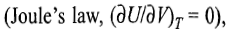
it follows that
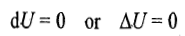
Substituting the above expression in the first law of thermodynamics, we get
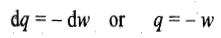
Hence in an isothermal expansion or compression process, heat is converted into work and vice versa. Thus, if heat is supplied to the system (q positive), the equivalent amount of work is done by the system (w negative), and if some work is done on the system (w positive), the equivalent amount of heat is given out (q negative). The enthalpy change of the system is also zero as
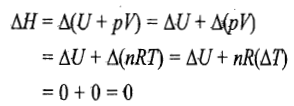
Reversible expansion or compression process
The expression for the work involved in an isothermal expansion or compression from volume V1 to V2 can be worked out as follows