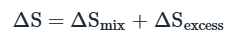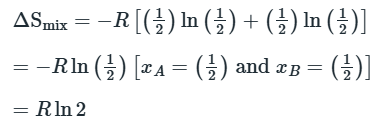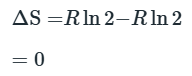Correct option is C
Let a number of gases at the same temperature and pressure be mixed as shown in Figure. Since the total volume after mixing is the sum of the individual volumes, the temperature and pressure of the system after mixing will be the same as that of unmixed gases. The free energy of mixing of the above process can be computed as follows.
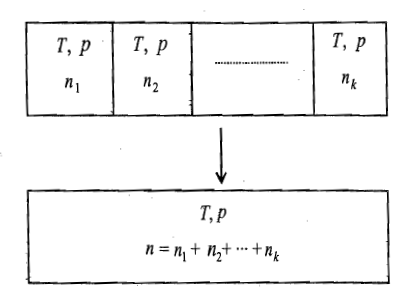
Free Energy of Unmixed Gases
Since each gas is present at the same T and P, its chemical potential is given by the expression
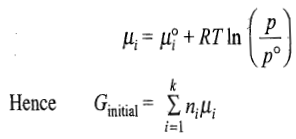
Free Energy of Mixed Gases
The pressure of the entire system is still P, but the individual gases have partial pressure given as
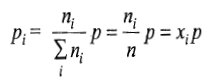
where xi is the amount fraction of the ith gas. The chemical potential of individual gases will be given by the expression
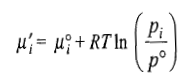
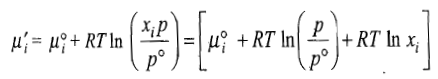
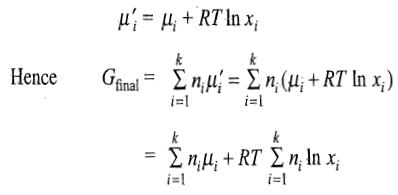
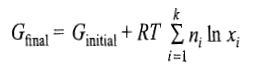
Free Energy of Mixing
The free energy of mixing is given as
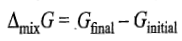
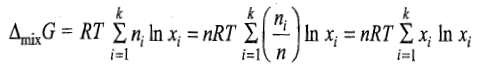
n represents the total amount of gases which are being mixed together.
Entropy of Mixing

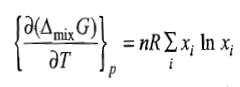
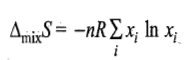
The negative sign means that ΔmixS is always positive.