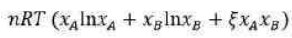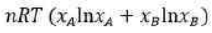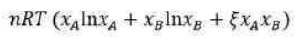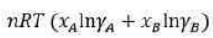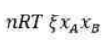Correct option is B
The Gibbs free energy (G) of a chemical system can be described using the Margules equation, particularly in the context of non-ideal solutions. The Margules equation is a thermodynamic expression that accounts for the deviations from ideal behaviour in solutions. It is typically used for binary solutions, where two components are mixed together.
Gibbs free energy when two components A and B are mixed to form binary solution
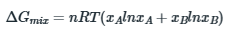
According to the Margules equation, after mixing of the components A and B, in order to determine strong interactions of A-B in comparison to A-A and B-B bond, coefficient
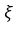
is used which is dimensionless.