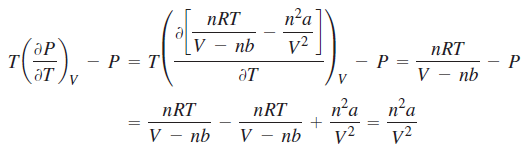Correct option is C
The van der Waals equation is a mathematical formula that describes the behavior of real gases. It is an equation of state that relates the pressure, volume, number of molecules, and temperature in a fluid.
One explicit way to write the van der Waals equation is:
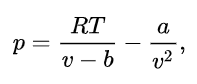
where is pressure, is temperature, and is molar volume, the ratio of volume, , to quantity of matter, is the Avogadro constant and the number of molecules). Also and are experimentally determinable, substance-specific constants, and is the universal gas constant. This form is useful for plotting isotherms (constant temperature curves).
Consider the dependence of U on V at constant T. This quantity has the units of
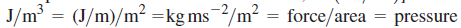
and is called the internal pressure.
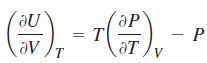
For a gas described by the van der Waals equation of state,