Correct option is D




A good trial wavefunction minimizes Etrial and closely approximates the actual ground state wavefunction.
For the Hamiltonian operator
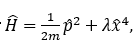
the best choice among the following trial variational wavefunctions for estimating the ground state energy is




A good trial wavefunction minimizes Etrial and closely approximates the actual ground state wavefunction.
The term symbol for the ground state of dinitrogen cation radical N2+is
For a model system of three non-interacting electrons confined in a two-dimensional square box of length L, the ground state energy in units of is
Consider a particle on a ring that is perturbed by interacting with an applied electric field (E) with the perturbation being is the dipole moment. The energy levels correct upto first order are
For an electron in 1s orbital of He+, the average value of is
Let the Hamiltonian H, in one-dimension, be
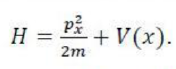
The commutator of H with x, [H,x], is
The equilibrium dissociation energy of a diatomic molecule is 4.75 eV and its stretching frequency corresponds to 0.5 eV. The minimum energy required to dissociate the molecule in eV is
The expectation value of p2 of a particle in a cubic box of side l, having the wave function
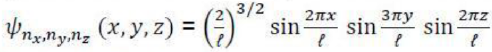
The quantum number corresponding to the z-component of the total electronic orbital angular momentum in the nitric oxide molecule is
When a hydrogen atom is exposed to a perturbation V=E.z, the first order correction to the wave function comes only from the orbital
The commutator,
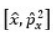
is equivalent to

