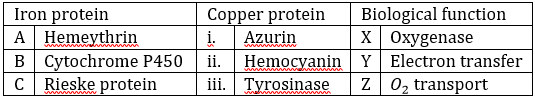
Correct option is D
Cytochromes are vital members of the mitochondrial electron-transfer chain. They are also essential components in plant chloroplasts for photosynthesis. Cytochromes are haem proteins, and the ability of the iron centre to undergo reversible
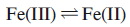
changes allows them to act as 1-electron transfer centres.
Many different cytochromes are known, with the reduction potential for the Fe3+/ Fe2+couple being tuned by the surrounding protein environment. Cytochromes belong to various families, e.g. cytochromes a, cytochromes b and cytochromes c, which are denoted according to the substituents on the haem group. In O2-carrying haem proteins, the ‘rest state’ contains a 5-coordinate Fe(II) centre which becomes 6-coordinate after O2 uptake. In contrast, the electron transfer cytochromes b and c contain 6-coordinate Fe which is present as either Fe(II) or Fe(III). There is little change in ligand conformation as the redox change occurs.
The structure of cytochrome c isolated from horse heart. In cytochrome c, the haem unit is bound to the protein backbone through axial His and Met residues, and through two Cys residues which are covalently linked to the porphyrin ring.
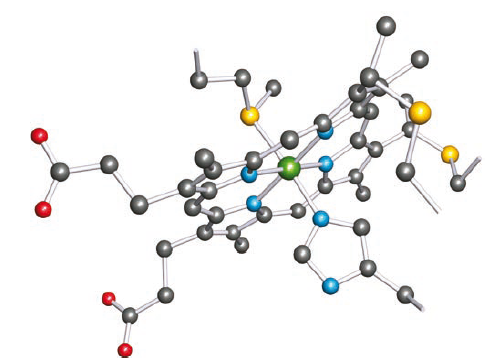
Hydrogen atoms have been omitted; colour code: Fe, green; S, yellow; N, blue; C, grey; O, red.
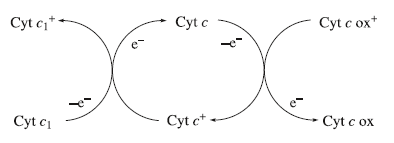
In the mitochondrial electron-transfer chain, cytochrome c accepts an electron from cytochrome c1 and then transfers it to cytochrome c oxidase. Ultimately, the electron is used in the 4-electron reduction of O2. The oxidized forms of the cytochromes contain Fe(III), and the reduced forms contain Fe(II).
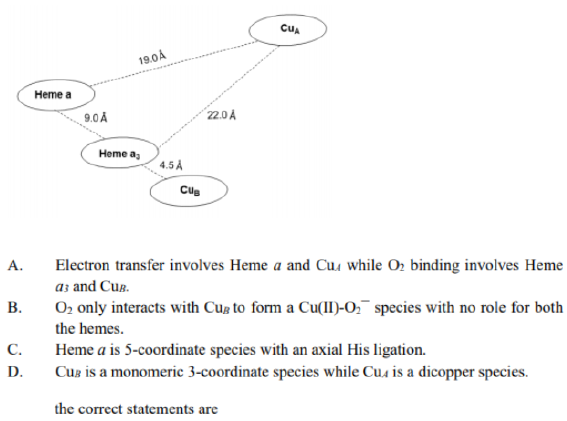
The exact nature of the metal sites in cytochrome c oxidase was resolved in 1995. This terminal member of the mitochondrial electron-transfer chain catalyses the reduction of O2 to H2O,and contains four active metal centres (CuA, CuB, haem a and haem a3) which couple electron transfer to proton pumping.
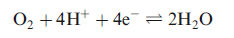
Electron transfer involves the CuA and haem a sites, electrons being transferred from cytochrome c to CuA and then to haem a. Haem a3 and CuB provide the site for O2 binding and O2 to H2O conversion, and are involved in pumping H+(four per O2 molecule) across the mitochondrial inner membrane.