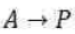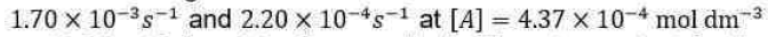Correct option is B
In a zero-order reaction, the rate of reaction
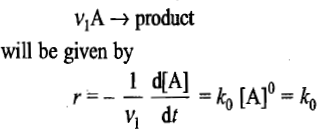
that is, the reaction proceeds at a constant rate and does not depend on the concentration of A.
Integrated Rate Law
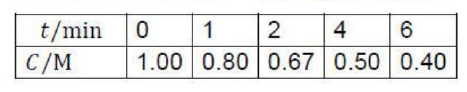
Let [A]0 be the concentration of A at t=0 and [A]t be the concentration at time t.

For the given reaction,
[A]=[A]0-kt
100% completion means [A]=0
t=[A]0/k