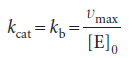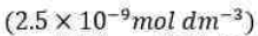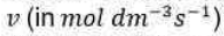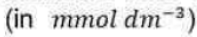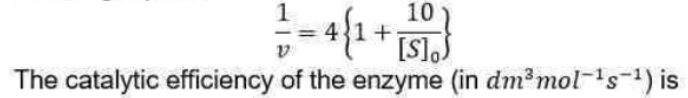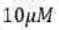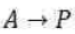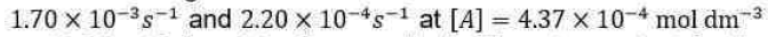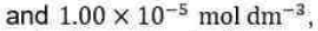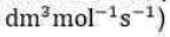Correct option is C
The Michaelis–Menten mechanism of enzyme catalysis
Experimental studies of enzyme kinetics are typically conducted by monitoring the initial rate of product formation in a solution in which the enzyme is present at very low concentration. Indeed, enzymes are such efficient catalysts that significant accelerations may be observed even when their concentration is more than three orders of magnitude smaller than that of the substrate. The principal features of many enzyme-catalysed reactions are as follows:

The Michaelis–Menten mechanism accounts for these features. According to this mechanism, an enzyme–substrate complex is formed in the first step and either the substrate is released unchanged or after modification to form products:
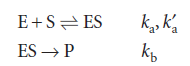
This mechanism leads to the Michaelis–Menten equation for the rate of product formation
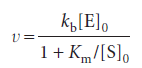

The rate of product formation according to the Michaelis–Menten mechanism is
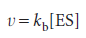
We can obtain the concentration of the enzyme–substrate complex by invoking the steady-state approximation and writing
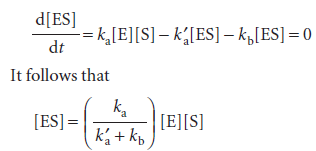

and note that KM has the same units as molar concentration. To express the rate law in terms of the concentrations of enzyme and substrate added, we note that [E]0=[E] + [ES]. Moreover, because the substrate is typically in large excess relative to the enzyme, the free substrate concentration is approximately equal to the initial substrate concentration and we can write [S] ≈ [S]0. It then follows that:
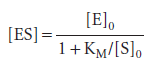
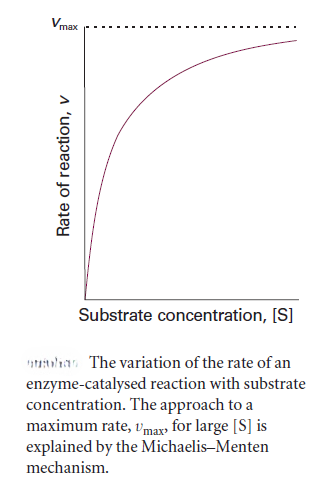

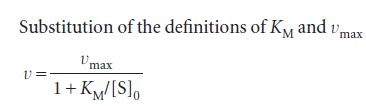
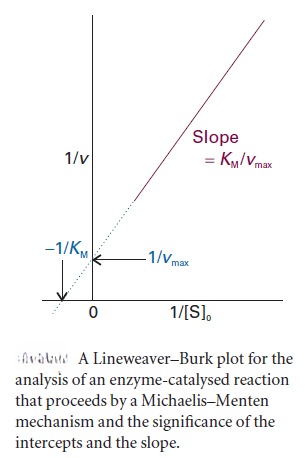
We can rearrange this expression into a form that is amenable to data analysis by linear regression:
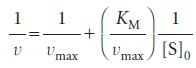
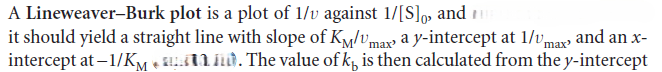
The catalytic efficiency of enzymes
The turnover frequency, or catalytic constant, of an enzyme, kcat, is the number of catalytic cycles (turnovers) performed by the active site in a given interval divided by the duration of the interval. This quantity has units of a first-order rate constant and, in terms of the Michaelis–Menten mechanism, is numerically equivalent to kb, the rate constant for release of product from the enzyme–substrate complex.