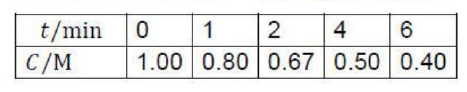
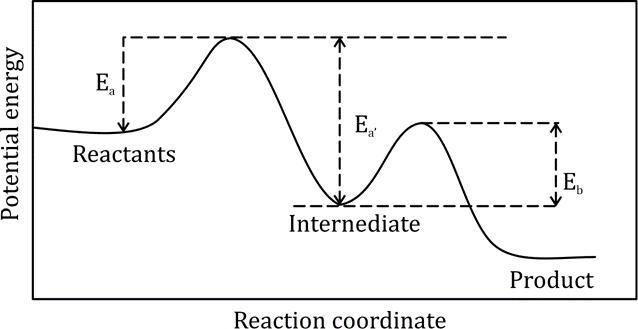
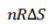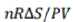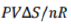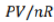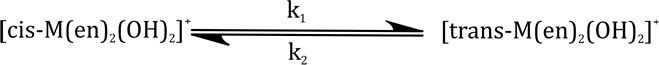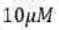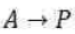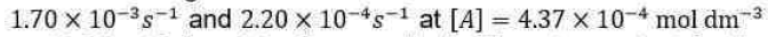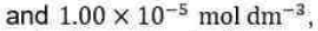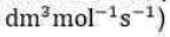Correct option is C
In an isothermal expansion process, the temperature of the system remains constant throughout the expansion process. Since for an ideal gas, U depends only on temperature
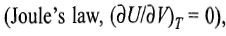
it follows that
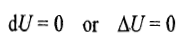
Substituting the above expression in the first law of thermodynamics (dU=dq+dw)
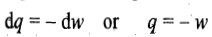
Hence in an isothermal expansion process, heat is converted into work and vice versa. Thus, if heat is supplied to the system (q positive), the equivalent amount of work is done by the system (w negative), and if some work is done on the system (w positive), the equivalent amount of heat is given out (q negative).

Reversible expansion process
The expression for the work involved in an isothermal expansion from volume V1 to V2 can be worked out as follows:

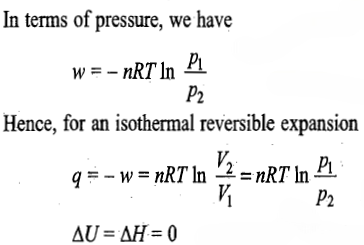

w=-q
w=-(-TΔS)=TΔS=(PV/nR)ΔS
[For an ideal gas, PV=nRT]