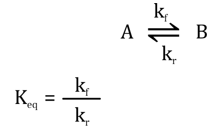Correct option is A
An enzyme catalyzes a reaction by lowering the activation energy, which increases the rate of the reaction. For the given reaction
<math xmlns="http://www.w3.org/1998/Math/MathML"><semantics><mrow><mi>A</mi><mo>↔</mo><mi>B</mi></mrow><annotation encoding="application/x-tex"> A \leftrightarrow B </annotation></semantics></math>A↔B, the forward rate (kf) and reverse rate (kr) define the equilibrium constant <math xmlns="http://www.w3.org/1998/Math/MathML"><semantics><mrow><msub><mi>K</mi><mrow><mi>e</mi><mi>q</mi></mrow></msub><mo>=</mo><mfrac><msub><mi>k</mi><mi>f</mi></msub><msub><mi>k</mi><mi>r</mi></msub></mfrac></mrow><annotation encoding="application/x-tex"> K_{eq} = \frac{k_f}{k_r} </annotation></semantics></math> Keq = kfkr\frac{k_f}{k_r}krkf
- Enzymes increase reaction rates: An enzyme will increase both <math xmlns="http://www.w3.org/1998/Math/MathML"><semantics><mrow><msub><mi>k</mi><mi>f</mi></msub></mrow><annotation encoding="application/x-tex"> k_f </annotation></semantics></math>kf (forward rate) and kr (reverse rate) by the same factor, as it lowers the activation energy for both directions of a reversible reaction.
- Effect on <math xmlns="http://www.w3.org/1998/Math/MathML"><semantics><mrow><msub><mi>K</mi><mrow><mi>e</mi><mi>q</mi></mrow></msub></mrow><annotation encoding="application/x-tex"> K_{eq} </annotation></semantics></math>Keq: Since Keq = , if both kf and kr increase by the same factor, their ratio (Keq) remains unchanged. Enzymes do not affect the equilibrium constant; they only speed up the rate at which equilibrium is reached.
- Effect on kr : The reverse rate Kr will increase due to the enzyme.
- Effect on : If kr increases, then will decrease.
- Effect on : Since keq remains unchanged,
also remains unchanged.
Now, let's evaluate the options:
1. kr: This is the reverse rate, which will increase due to the enzyme. This matches our analysis.
2. keq : The equilibrium constant does not change with an enzyme, so this is incorrect.
3. : Since kr increases, <math xmlns="http://www.w3.org/1998/Math/MathML"><semantics><mrow><mn>1</mn><mi mathvariant="normal">/</mi><msub><mi>k</mi><mi>r</mi></msub></mrow><annotation encoding="application/x-tex"> 1/k_r </annotation></semantics></math>1/kr decreases, so this is incorrect.
4. : Since keq remains unchanged, also remains unchanged, so this is incorrect.
Answer: The parameter that will be increased by the enzyme over the uncatalyzed reaction is:
1. kr