Correct option is A
Explanation-
Uncompetitive inhibitors bind only to the enzyme-substrate complex (ES), not to the free enzyme.
This binding typically lowers both Vmax and Km.
1. Vmax decreases because the inhibitor prevents the enzyme from converting substrate to product.
2. Km also decreases because the formation of the ES-inhibitor complex stabilizes the ES complex, effectively increasing the enzyme's affinity for the substrate.
Statement A: "Uncompetitive inhibitor binds to both free enzyme as well as an enzyme-substrate complex." - Incorrect
Uncompetitive inhibitors bind only to the enzyme-substrate complex (ES) and not to the free enzyme (E). Binding occurs at a site other than the active site and only after the substrate is bound.
The defining feature of uncompetitive inhibition is exclusive binding to ES, which alters the enzyme activity without competing with substrate at the active site.
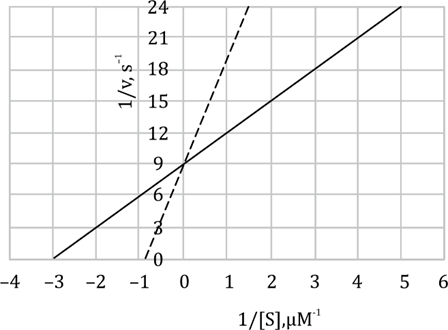
Statement B: "Addition of uncompetitive inhibitor lowers the Vmax of the reaction." - Correct
Vmax is the maximum rate of the reaction when the enzyme is saturated with substrate. In uncompetitive inhibition the inhibitor binds to the ES complex and prevents the complex from forming product. As a result, even if we add more substrate, you cannot achieve the original Vmax, because some ES complexes are "trapped" in inactive EI(S) complexes. Since fewer ES complexes can proceed to product, Vmax is lowered.
Statement C: "Apparent Km of the enzyme is lowered." - Correct
Km (Michaelis constant) is a measure of the enzyme’s affinity for its substrate. In uncompetitive inhibition the inhibitor binds to ES complex, stabilizing it. This leads to a reduction in the apparent Km, since the ES complex is stabilized and dissociation back to free enzyme and substrate is less likely. This is observed as a lower Km, which indicates increased apparent substrate affinity, although it’s due to ES-inhibitor complex formation.
Statement D: "Apparent Km of the enzyme remains unchanged." - Incorrect
As explained above, apparent Km is lowered in uncompetitive inhibition. This statement would be true for noncompetitive inhibition, where Km often remains unchanged (in pure noncompetitive cases).
So, the correct answer is option a - B and C are correct statements.