Correct option is B
Explanation-
To solve this, we use the Michaelis-Menten equation with competitive inhibition:
Given-
Explanation-
To solve this, we use the Michaelis-Menten equation with competitive inhibition:
Given-
What is the fold difference between v at [S] = Km and v at [S] = 1000Km, where v is the initial velocity of an enzyme catalyzed reaction, [S] is substrate concentration and Km is the Michaelis constant?
The following statements are being made to define the Michaelis constant (Km). It is:
A. Independent of enzyme concentration [E] and substrate concentration [S]
B. Equal to the dissociation constant when the [ES] complex dissociates more rapidly than product formation
C. Equal to the dissociation constant when product formation is more rapid than [ES] complex dissociation
D. An intrinsic property of an enzyme and does not depend on pH, temperature, and ionic strength
Which one of the following combination of statements is correct?
The enzyme alkaline phosphatase was tested for its catalytic activity using the substrate para-nitrophenylphosphate. The obtained was 10 mM and was 100 µmol/min. Which one of the following options represents the initial velocity of the reaction at a substrate concentration of 10 mM?
Following statements are made about uncompetitive inhibition of an enzyme:
A. Uncompetitive inhibitor binds to both free enzyme as well as an enzyme-substrate complex.
B. Addition of uncompetitive inhibitor lowers the Vmax of the reaction.
C. Apparent of the enzyme is lowered.
D. Apparent of the enzyme remains unchanged.
Which one of the following options represents the correct combination of the statements?
Given below are plots of the linear derivation of Michaelis-Menten kinetic equation and statements related to the variables: initial velocity (V₀) and substrate concentration [S].
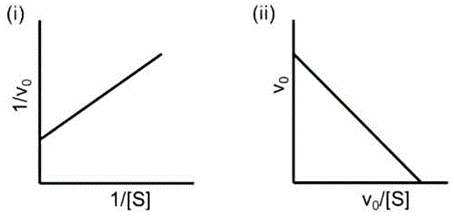
A. In plot (i), both x and y axes have dependent variables.
B. In plot (ii), neither x nor y axis has independent variables.
C. In plot (i), only y-axis has a dependent variable.
D. In both the plots, x-axis has an independent variable.
Select the options that has all the correct statements?
A student was asked to plot a graph representing enzyme kinetic data for initial velocity, v₀, and substrate concentration, [S] using any of the equations given below. The student used an equation for which neither X-axis nor Y-axis had independent variables. Which one of the following equations might the student have used?
Dixon plot is used to study the enzyme inhibition by plotting various expressions of velocity (v) and inhibitor concentration [I] on the X-axis (Column A) and Y-axis (Column B) as given below:
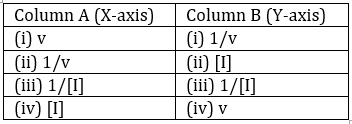
Which one of the following options is the correct combination from columns A and B to draw the Dixon plot?
The figure below depicts the allosteric regulation in the biosynthesis of three aromatic amino acids - Phe, Tyr and Trp, acting at four major steps catalyzed by enzymes, CM, AS, ADT and ADH. The feedback regulation and relaxation of enzyme activities by the end-product amino acids are marked.
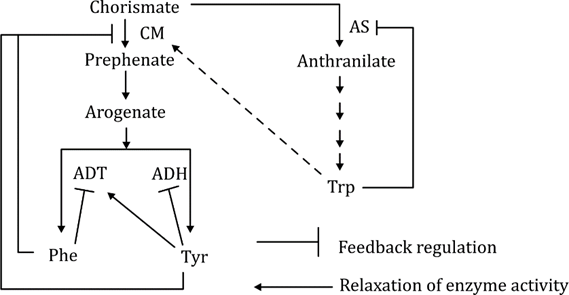
Following assumptions are made regarding the pool of aromatic amino acids in the feedback-insensitive mutants of these allosteric enzymes.
A. The feedback-insensitive mutant of CM will show higher pool of Phe and Tyr.
B. The feedback-insensitive mutant of AS will increase only Trp pool.
C. The feedback-insensitive mutant of AS will show higher pool of Trp, Phe and Tyr.
D. In feedback-insensitive mutant of ADH, only Tyr pool is decreased.
E. In feedback-insensitive mutant of ADH, both Tyr and Phe pools are increased transiently.
Which one of the following options represents a combination of all correct assumptions?
The graph below shows the plot of 1/v vs 1/[S] for an enzymatic reaction, with the solid and dashed lines representing the reactions without and with an inhibitor, respectively. The concentration of the inhibitor is 1 μM.
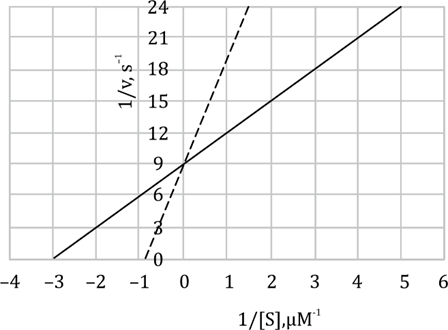
Which one of the following will be theof the inhibitor?

