Correct option is A
Axial attack is preferred with unsaturated six-membered rings
Six-membered rings with one trigonal (sp2) carbon atom can undergo axial or equatorial attack.
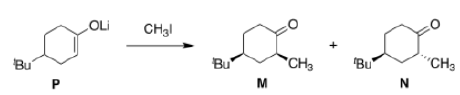
Six-membered rings with two or more trigonal carbon atoms undergo axial attack in order to form chairs rather than boats. The final product may end up with axial or equatorial substitution, but this is not a consideration in the reaction itself.
Alkylations of enolates, enamines, and silyl enol ethers of cyclohexanone usually show substantial preference for axial attack. The enamine of 4-t-butylcyclohexanone, which has a fixed conformation because of the t-butyl group, gives 90% axial alkylation and only 10% equatorial alkylation with n-PrI.
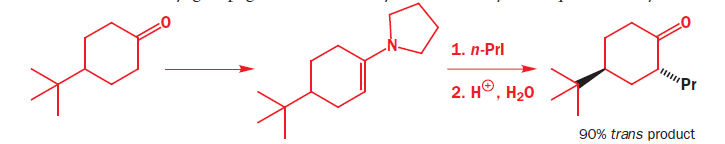
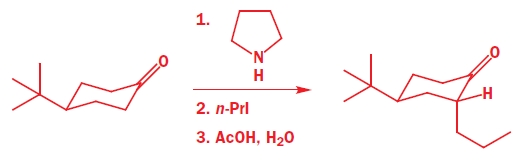
It is a simple matter to show that the preferred product has the new propyl group in the axial position because both the starting ketone and the product have chair conformations with the t-butyl equatorial. To get at the explanation we need to look at the conformation of the enamine intermediate. At this point we shall generalize a bit more and write a structure that represents any enol derivative where X may be OH, O- , OSiMe3 , NR2 , and so on. The conformation has a double bond in the ring, and is a partially flattened chair.
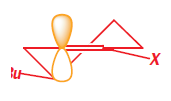
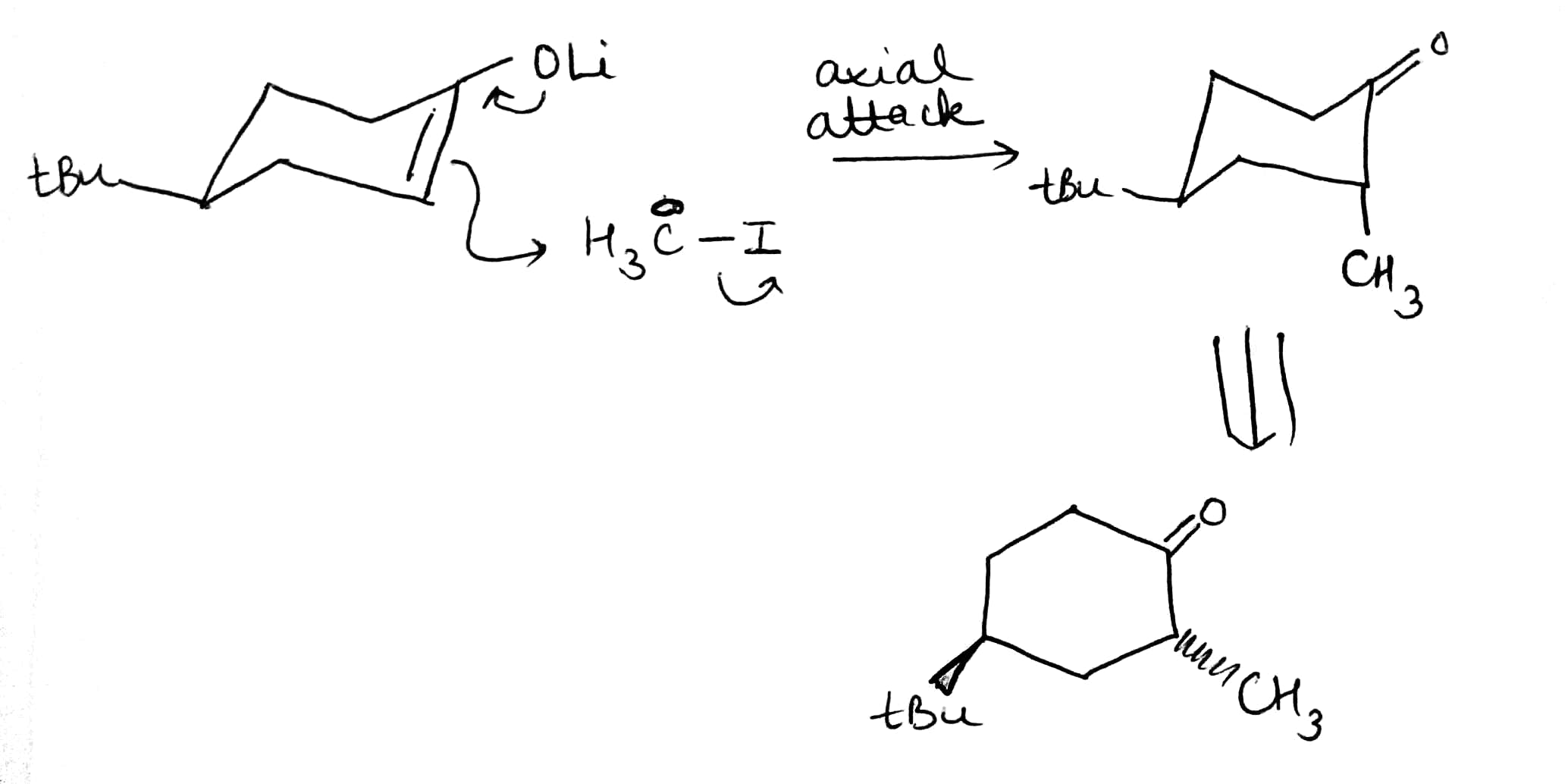
The t-butyl group is in an equatorial position at the back of the ring. The electrophile must approach the enol derivative from more or less directly above or below because only then can it attack one of the lobes of the p orbital at the enol position shown in yellow. The top of the molecule looks to be more open to attack so we shall try that approach first.
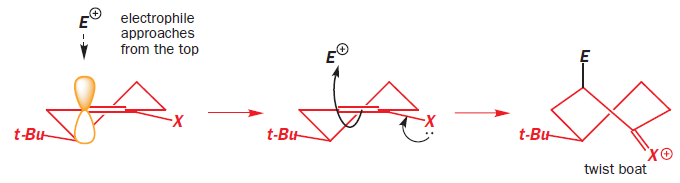
As the electrophile bonds to the trigonal carbon atom, that atom must become tetrahedral and it does so by forming a vertical bond upwards. The result is shown in the diagram—the ring turns into a twist-boat conformation. Now, of course, after the reaction is over, the ring can flip into a chair conformation and the new substituent will then be equatorial, but that information is not present in the transition state for the reaction. We could say that, at the time of reaction, the molecule doesn’t ‘know’ it can later be better off and get the substituent equatorial: all it sees is the formation of an unstable twist boat with a high-energy transition state leading to it.
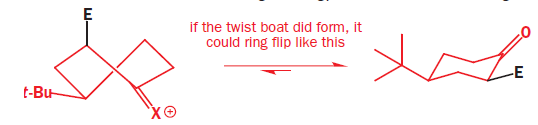
Attack from the apparently more hindered bottom face makes the trigonal carbon atom turn tetrahedral in the opposite sense by forming a vertical bond to the electrophile downwards. The ring goes directly to a chair form with the electrophile in the axial position.
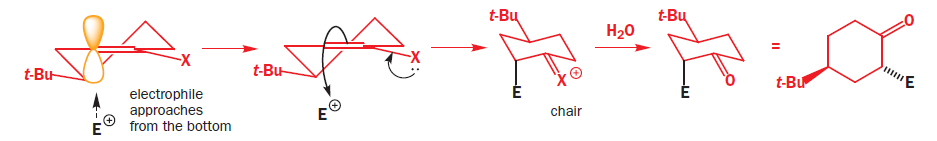
When the carbonyl group is restored by hydrolysis (if necessary-X may be O already) the ring need not flip: it’s already a chair with the t-butyl equatorial, and the new substituent is axial on the chair. This is the observed product of the reaction.
The reagent has to attack from an axial direction to interact with the p orbital. If it attacks from above, the new substituent is axial on an unstable twist boat. If it attacks from below, the new substituent is axial on a chair— granted, this is not as good as equatorial on a chair, but that’s not an option—it has to be axial on something, and a chair is better than a twist boat. So this is the product that forms. It’s just hard luck for the substituent that it can’t know that if it did weather it out on the twist boat it could later get equatorial—it plumps for life on the easy chair and so has to be content with ending up axial.