Correct option is D
In second order reactions, the rate expression depends on the two concentration terms. These concentration terms may refer to either the same species or different species. Thus, two cases may be distinguished. These are:


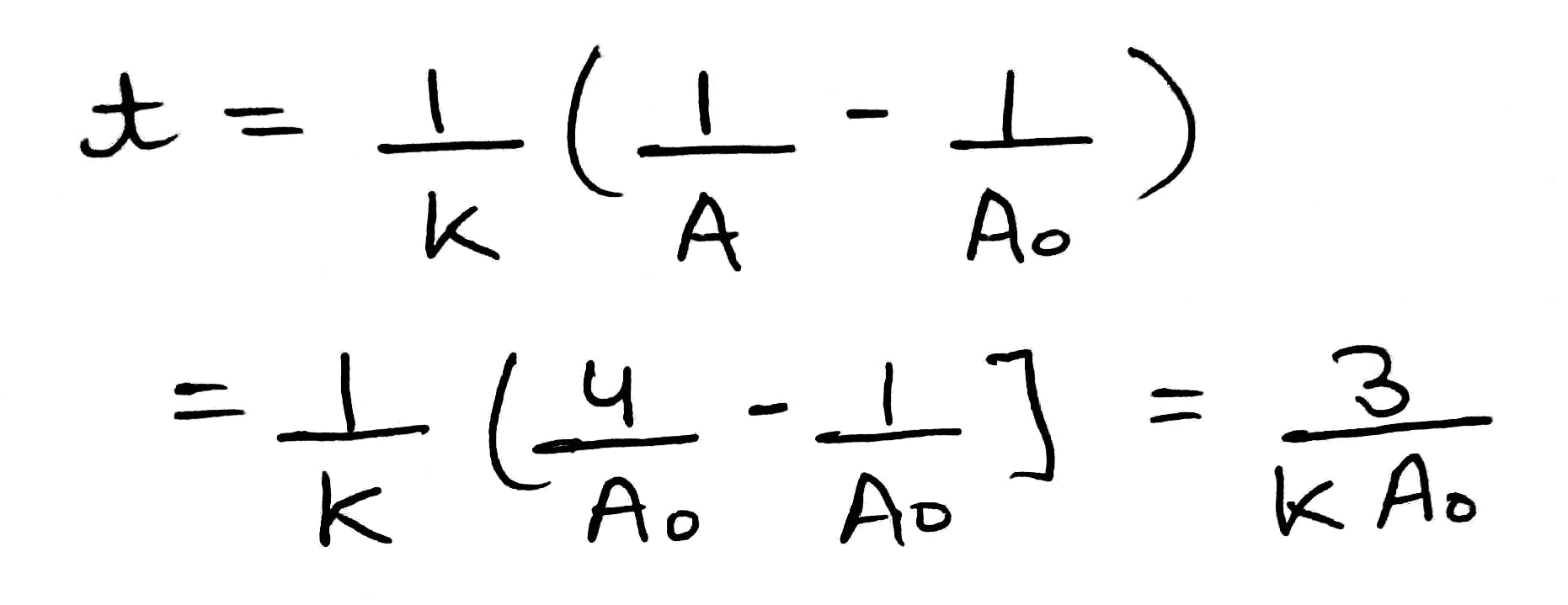
A reaction follows the rate law
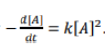
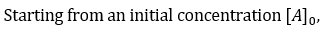
the time taken for the concentration to reduce to

is
In second order reactions, the rate expression depends on the two concentration terms. These concentration terms may refer to either the same species or different species. Thus, two cases may be distinguished. These are:


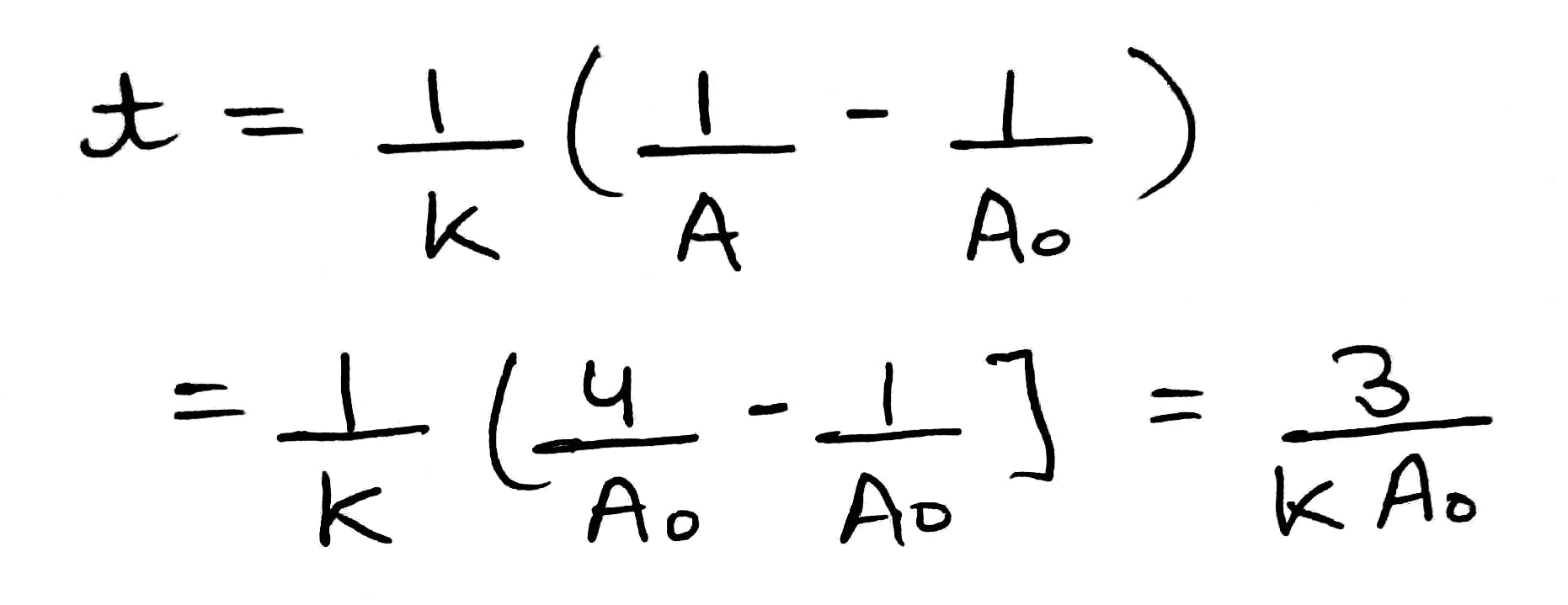
Substance ‘A’ is exposed to 600nm, 100 W light source for 6626s, with 50% of the incident light being absorbed. ‘A’ decomposes according to the reaction A→B . As a result of irradiation, 0.2 mol of B is produced. The quantum yield of the reaction is closest to
The concentration (C) of a compound undergoing decomposition as a function of time (t) is given below:
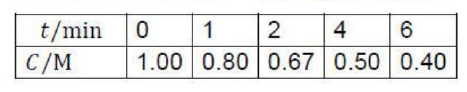
The order of the reaction is
For a reaction, raising the temperature from 200K to 300K results in the increase of rate constant by a factor of 2. The activation energy for this reaction in kJ mol-1 is closest to (ln2=0.69).
For the reaction,
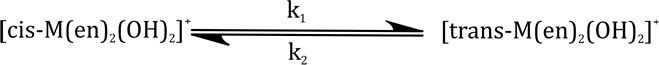
the equilibrium constant is 0.16 and k1 is 3.3×10-4s-1.The experiment is started with pure cis from. The time taken for half the equilibrium amount of trans isomer to be formed is about
The rate constant for the reaction, A2B4O→AB4 +AO is described as, . The activation energy for this reaction (in kJ mol-1) is closest to
The rate of decomposition of a gas is The order of the reaction is:
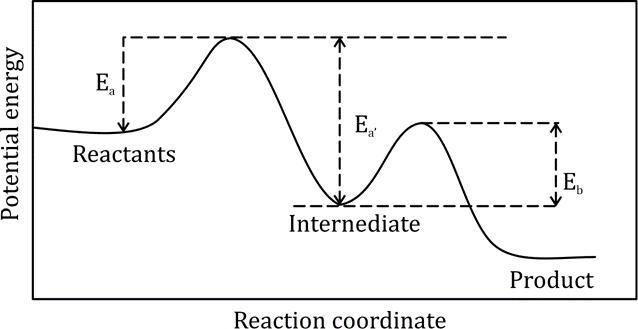
The effective activation energy for the reaction:
with the following potential energy versus reaction coordinate plot is
A protein has 3 tyrosine residues and ‘n’ tryptophan residues both of which are the only amino acids absorbing at 280nm. If the absorbance of the protein having a concentration of
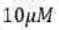
in a cuvette of path length 2 cm) is 0.59, the number of tryptophan residues in the protein must be

The effective rate constants for a gaseous unimolecular reaction:
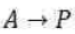
following the Lindemann-Hinshelwood mechanism are
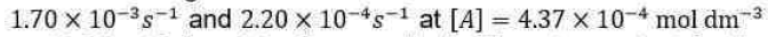
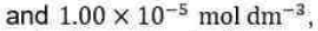
respectively. The rate constant for the activation step in the mechanism is approximately equal to (in
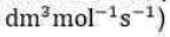
The predicted rate law, using the steady state approximation, for the reaction


