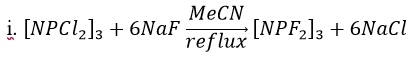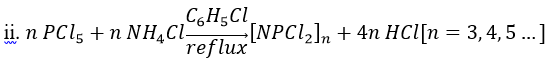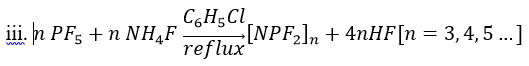Correct option is D
Phosphazenes refer to various classes of organophosphorus compounds featuring phosphorus(V) with a double bond between P and N. One class of phosphazenes have the formula R−N=P(−NR2)3. These phosphazenes are also known as iminophosphoranes and phosphine imides. They are superbases.
Hexachlorophosphazene is an inorganic compound with the chemical formula (NPCl2)3. The molecule has a cyclic, unsaturated backbone consisting of alternating phosphorus and nitrogen atoms, and can be viewed as a trimer of the hypothetical compound N≡PCl2 (phosphazyl dichloride). Its classification as a phosphazene highlights its relationship to benzene.
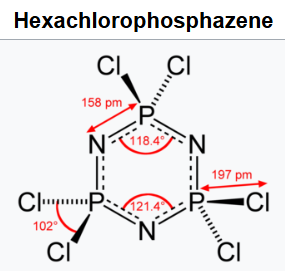
The synthesis of hexachlorophosphazene was first reported by von Liebig in 1834. It was found that phosphorus pentachloride (PCl5) and ammonia (NH3) react exothermically to yield a new substance that could be washed with cold water to remove the ammonium chloride
([NH4]Cl) coproduct. The new compound contained P, N, and Cl, on the basis of elemental analysis. It was sensitive toward hydrolysis by hot water.
By replacing ammonia with ammonium chloride allows the reaction to proceed without a strong exothermic reaction associated with the NH3/PCl5. Typical chlorocarbon solvents are 1,1,2,2-tetrachloroethane or chlorobenzene, which tolerate the hydrogen chloride (HCl) side product. Since ammonium chloride is insoluble in chlorinated solvents, workup is facilitated. For the reaction under such conditions, the following stoichiometry applies:
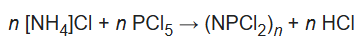
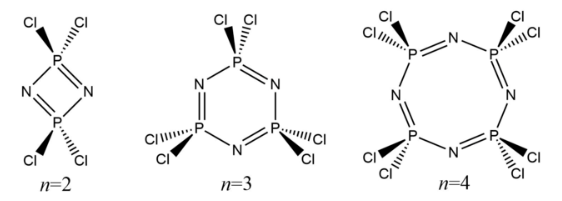
where n can usually take values of 2 (the dimer tetrachlorodiphosphazene), 3 (the trimer hexachlorotriphosphazene), and 4 (the tetramer octachlorotetraphosphazene).
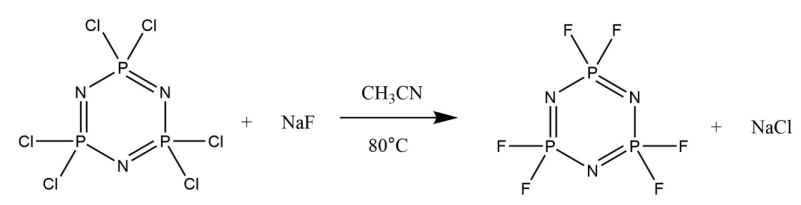
Hexafluorocyclotriphosphazene: Due to the chlorotrimer’s importance in past phosphazene reactions, finding a synthetic route that consistently produced hexafluorocyclotriphosphazene ([NPF2]3) at a high purity was extremely important. A number of pathways were reported in the literature with various fluorinating agents or catalysts. This procedure was selected as it involved only two reactants, [NPCl2]3 and NaF, meaning it would likely be the simplest way to get the high purity product that was desired.